Understanding the Complicated Relationship Between Insulin, Glucagon and GIP
The Revolutionary Discoveries of a Diabetic Scientist
By Johanna Lee • November 2021
Dr. Kimberley El has always loved science. In school, she enjoyed chemistry and math, and initially she wanted to be an analytical chemist. But in her junior year of college, life threw her a curve ball. She was diagnosed with type 1 diabetes, an autoimmune disease in which the insulin-producing β cells in the pancreas are destroyed and eventually stop producing insulin. “It’s just this one little cell, and it changed my whole life,” she says.
Like all patients with type 1 diabetes, she relies on daily injections of insulin to survive. Despite struggling with her illness every day, she did not lose her passion for science. Her disease inspired her to change course and pursue a graduate degree in biomedical science. She earned her PhD in Cellular Physiology from the Indiana University School of Medicine and later joined the lab of Dr. Jonathan Campbell in the Department of Medicine at Duke University as a post-doctoral fellow. Her research focuses on understanding how insulin and glucagon regulate energy metabolism—the very system that has gone awry in her own body. Specifically, she studies a phenomenon called “the incretin effect”.

The Incretin Effect
When glucose is given to someone via intravenous injection, their blood glucose will increase, and then slowly decrease. However, when glucose is given orally, the blood glucose levels go down much faster. The reason is that the gut can detect nutrients and stimulate the release of certain factors (“incretins”) that signal the pancreas to secrete insulin and help regulate glucose levels. This is known as “the incretin effect”. One of the most important incretins is the glucose-dependent insulinotropic polypeptide (GIP).
The pancreas consists of various types of islet cells, including α cells and β cells. What has been taught in biology classes for decades is that α cells secrete glucagon to raise glucose levels, and β cells secrete insulin to lower glucose. Glucagon and insulin have long been portrayed as enemies, fighting against each other. GIP is released from the gut when high glucose is detected and binds to the GIP receptor on β cells, which then stimulates insulin secretion to lower blood glucose. But GIP also stimulates glucagon secretion. In fact, GIP receptors are expressed in both α cells and β cells. So if insulin and glucagon work in opposition, why would GIP induce both insulin and glucagon secretion? There seemed to be a missing piece of the puzzle, and Dr. El was determined to find it.
The Dual Actions of GIP
Dr. El began investigating this missing piece by looking into the ideal environment in which GIP stimulates glucagon secretion. Most of the previous studies on the incretin effect focused on the role of glucose. In the real world, however, our meals consist of a variety of nutrients besides carbohydrates, such as proteins that break down into amino acids.
She hypothesized that amino acids may be the nutrient “fuel” for α cells, the same way that glucose is the fuel for β cells. Her results showed that there is, in fact, a synergy between GIP and amino acids. “When we give GIP, we get a little bit of glucagon. When we give amino acids, we also get a little bit of glucagon. But when we give both GIP and amino acids together, we get a whole ton of glucagon,” she says. This was the missing piece that changed our understanding of the role of GIP and the relationship between glucagon and insulin.
It turns out that GIP stimulates insulin secretion through dual actions: 1) In the presence of glucose, GIP directly stimulates β cells to secrete insulin; and 2) In the presence of amino acids, GIP stimulates α cells to induce glucagon secretion, which sends a message to β cells to induce insulin secretion.
“In the past, people viewed this relationship as insulin drives glucose down and glucagon drives glucose up, and they kind of fight each other. But we're finding that they actually talk to each other,” she says. “In fact, there's actually glucagon receptors on the β cell. And previous studies in our lab have shown that glucagon talks to the β cell to increase insulin secretion. So they're not exactly in opposition all the time; we’re seeing that there is a lot more crosstalk between cells. This is so important for maintaining proper levels of these islet hormones to regulate blood glucose in that tight, tight window. It's so fine-tuned in a healthy person—it’s amazing!”
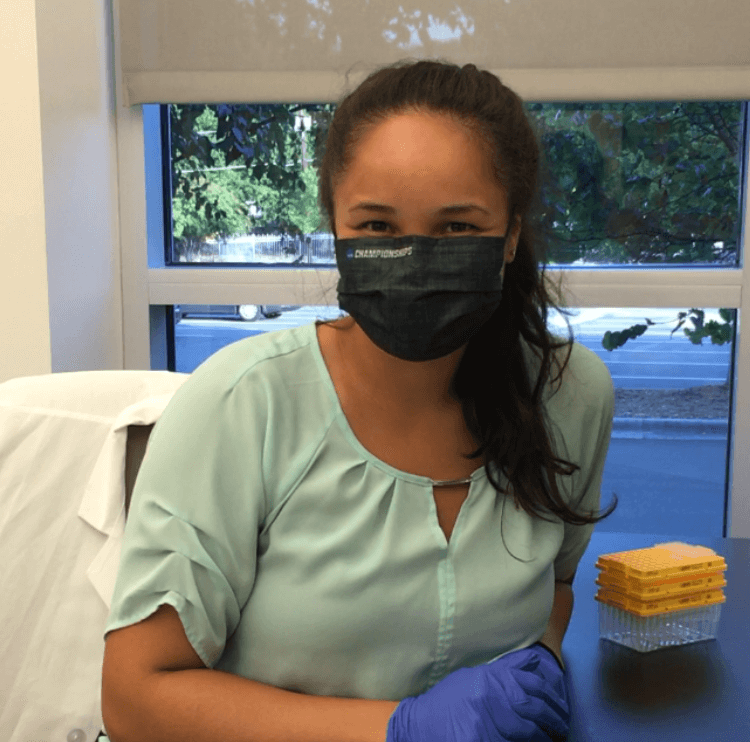
Methods for Studying Islet Biology
Studying islet biology is challenging because islet cells are constantly responding to changing stimuli, and changes happen quickly. In order to capture these responses in a timely manner, Dr. El used a method called “perifusion”. The islets are placed in chambers and solutions containing stimuli are passed through them. The islets respond by secreting hormones such as glucagon or insulin, and the secreted samples are collected minute-by-minute.
Collecting samples, however, is only the first step. In Dr. El’s case, samples are collected over a long time-course, and she can generate up to 1,200 samples in just one experiment! The next challenge is to analyze this massive number of samples. Traditionally, glucagon levels are measured using ELISA, which is not only expensive, but also takes a long time. Labs either have to spend a lot of time and money assaying all the samples, or assay fewer time points and lose resolution—both of which are not ideal.
Dr. El was able to solve this issue using the new Lumit™ Glucagon Immunoassay. The assay does not require any wash steps and can be done in a 384-well plate. “With the Lumit™ Assay, we can measure every sample, and we can do it pretty quickly. The ELISA is an overnight assay, while the Lumit™ Assay I can run in 2 hours. That’s a 16-hour difference! It's a lot of time in which I could be doing other things,” she says. “If I’m busy, I can do perifusions twice a week. To have the data before I move on to my next experiment is very useful to me.”
The Future of Glucagon
For decades, glucagon has been seen as the “bad guy”. People assume that because diabetic patients have high glucagon levels, that is what is causing the disease. Because of this, any drug targets that may increase glucagon, such as GIP, were passed over as potential targets for treating diabetes. However, Dr. El’s research suggests that the high glucagon levels may not be the cause of disease. Instead, the islet cells may be compensating for low insulin by inducing more glucagon secretion. “This study shows that glucagon isn't so bad. And just because something induces glucagon, it doesn't mean it can't be a target for therapeutics,” she says.
In the future, Dr. El wants to determine if agents that induce glucagon secretion can be used to counter hypoglycemia in patients, such as herself, who have to self-administer insulin and are at higher risk of episodes of insulin overdose and severe low blood glucose. Her research could potentially lead to new therapeutic approaches for treating diabetes and benefit millions of patients around the world. When looking back at her decision to choose biomedical research, she says, “I think it was a really good change for me. It's interesting and relevant to the real world. It’s easy to see how my research affects a person, which I find really fulfilling. It's such a struggle for anybody with diabetes. It's a daily, hourly thing—it's always there. So, finding a way to alleviate any part of that stress is what motivates me.”
Reference: El, K. et al. (2021) GIP mediates the incretin effect and glucose tolerance by dual actions on α cells and β cells. Sci Adv. 7, eabf1948.
Interested in resources for Diabetes Research?
Explore webinars, posters, products and more on our Diabetes Research page.
Related Resources
Webinar: Islet Microtissues for Diabetes Research
Learn how 3D islet microtissues and 3D-validated assays can be used in diabetes research.
Immunoassays to Detect Metabolic Regulators
Learn about no-wash Lumit™ Immunoassays for fast, easy measurement of insulin and glucagon.

Homogeneous Assays For Triglyceride Metabolism Research
Learn about simple triglyceride and glycerol assays that can monitor the effect of insulin on lipid metabolism.