Sequencing Primers for Flexi® Vector Inserts
Sarah Wheeler, Jim Hartnett, Ethan Strauss, and Trista Schagat
Promega Corporation
Publication Date: December 2013
Abstract
When first cloning PCR-amplified inserts into the Flexi® Vector System, it is necessary to sequence the construct to confirm the presence of the desired clone. Transfer between Flexi® Vectors uses a high-fidelity method and does not require resequencing. This article lists primer sequences that can be used to sequence Flexi® Vector inserts.
Introduction
The Flexi® Vector System is a simple, yet powerful, directional cloning method for protein-coding sequences. It is based on two rare-cutting restriction enzymes, SgfI and PmeI, and provides a rapid, efficient and high-fidelity way to transfer protein-coding regions between a variety of Flexi® Vectors without the need to resequence.
When first creating a Flexi® Vector construct, the protein-coding region is amplified using primers that append SgfI and PmeI restriction sites on either end of the sequence. These restriction enzyme sites are then used to clone the PCR product into the Flexi® Vector. Even when using thermostable DNA polymerases with proofreading activity, such as Pfu DNA polymerase, it is still necessary to sequence 1–2 clones to ensure no errors were introduced during the PCR. Taq DNA polymerase is not recommended for cloning protein-coding regions due to the relatively high error rate, but if this polymerase is used, 5–6 clones will typically need to be sequenced to find a 2 kilobase insert that is error-free.
Flexi® Vector Sequencing Primers
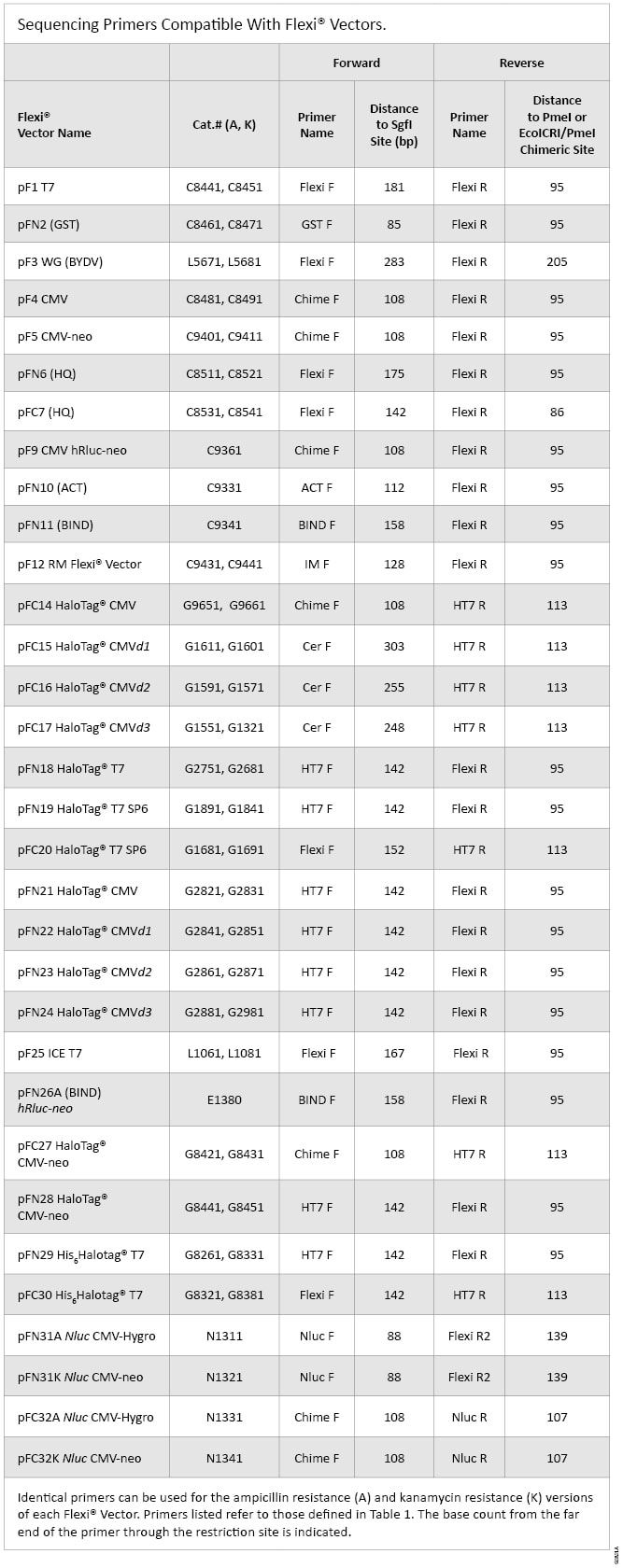
Listed in Table 1 are the sequencing primers used by Promega scientists to confirm insert sequences in the Flexi® Vectors. Table 2 is a complete list of the Flexi® Vectors and which sequencing primers have been used to sequence inserts cloned into each vector.
Table 1. Sequencing Primers for Flexi® Vector Inserts.
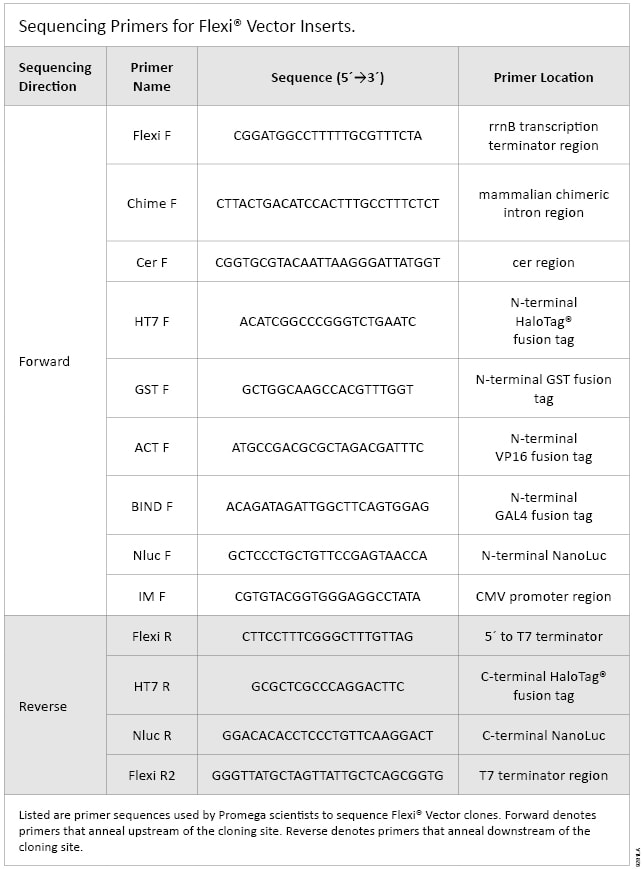
Table 2. Sequencing Primers Compatible with Flexi® Vectors.
Summary
When first cloning a protein-coding region into the Flexi® Vector System, it is necessary to sequence the insert to ensure no errors were introduced during PCR. Subsequent transfers of the protein-coding region into other Flexi® Vectors use a high-fidelity transfer method, so resequencing the coding region is not necessary. The primers listed here have been used to sequence inserts in each of the indicated Flexi® Vectors.
Learn more about the Flexi® Cloning System