Target Cell Killing Bioassays
Our HiBiT Target Cell Killing (TCK) Bioassays provide a simple, sensitive and highly specific method to measure target cell killing induced by a variety of biologic drugs. Upon lysis, the assays generate a bright luminescent signal that can be measured using a standard luminometer.
How Do TCK Bioassays Work?
Faster Processing Times, Dynamic Signal-to-Noise Ratio and Easy-to-Interpret Data
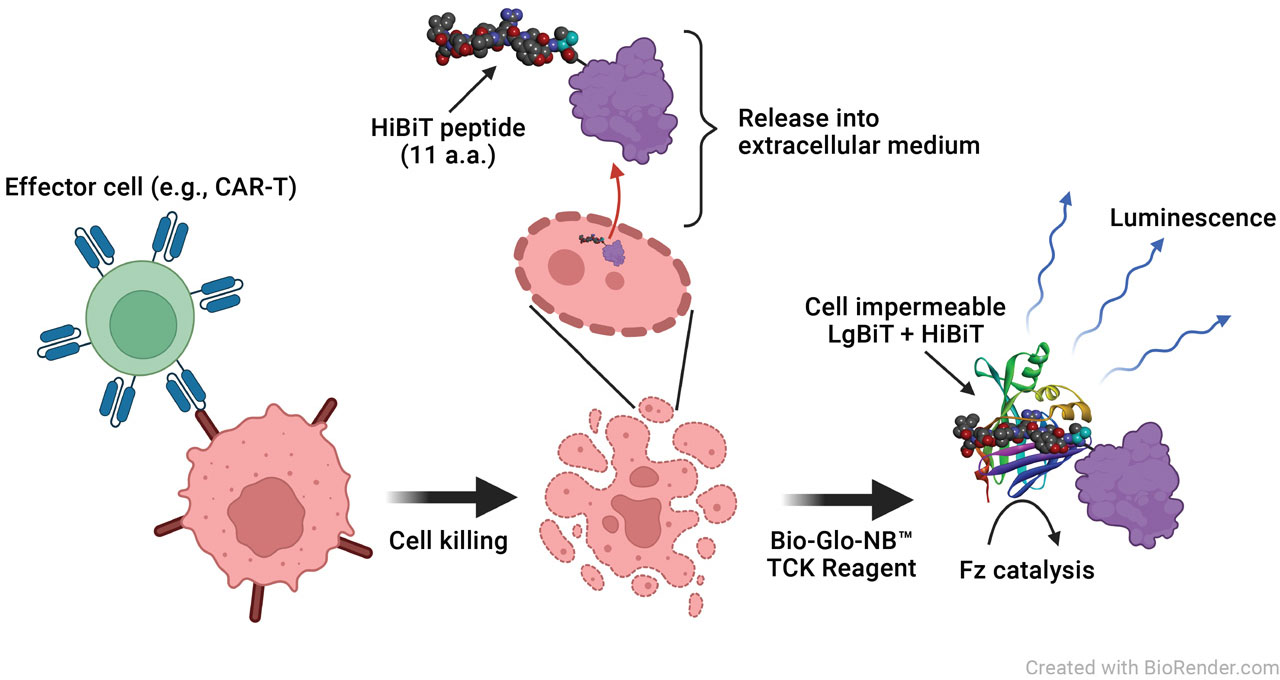
Principle of the HiBiT TCK Bioassay. Cytotoxic mAbs and/or effector cells are incubated with target cells expressing a HiBiT fusion protein. Upon killing of the target cell, the HiBiT fusion protein is released and binds extracellular LgBiT to create a functional NanoBiT® Luciferase enzyme. Luminescence is measured using a luciferase substrate and the GloMax® Discover Microplate Reader.
TCK Cell Lines for Immuno-Oncology Research
Primary Effector Cells
PBMCs
- ADCC-qualified
- Single-donor derived
- FcγR genotyped
CD8+ T cells
- TDCC qualified
- Functionally tested
Macrophage
- ADCP qualified
- Functionally tested
- FcγR genotyped
TCK Cell Line Screening
- ViaScript® (HiBiT) TCK Bioassay
- Transient expression of HiBiT
- Identify optimal TCK line
- For blood cancer or solid tumor cell lines
Solid Tumor Target Cells
Ovarian Carcinoma
- OVCAR3 and SKOV3
- Expressing: HER2, MSLN, 5T4, MUC16
- MSLN-KO line available
Breast Adenocarcinoma
- SK-BR-3
- Expressing: HER2, EpCAM
Lung Carcinoma
- A549
- Expressing: EGFR
Melanoma
- A375
- Expressing: HER2, CD70, B7-H3
Blood Cancer Target Cells
B cell Lymphoma and Leukemia
- Raji (HaloTag-HiBiT)
- Raji (LDH-HiBiT)*
- Ramos
- Cell lines expressing CD19, CD20, CD22 and CD38
- Knockout lines available for some targets
Myeloid Leukemia
- U937 and K562
- Expressing: CD33 and CLL-1
Multiple Myeloma
- H929
- Expressing: BCMA and CD38
T cell Leukemia
- T2
- Expressing: CD5, CD7, CD30, and CD52
*No functional differences have been observed between LDH and HaloTag® cell lines.
Have you identified an optimal cell line with ViaScript® (HiBiT) TCK Bioassay?
Our Tailored R&D Solutions (TRS) Team builds clonal TCK lines.
Applications
The HiBiT TCK platform can be used to measure TCK activity induced by a variety of biologic drug modalities. The data included here
are representative of three applications: CAR-T cell-mediated cytotoxicity, antibody-dependent cell-mediated cytotoxicity (ADCC),
T cell-dependent cell-mediated cytotoxicity (TDCC) and antibody-dependent cellular phagocytosis (ADCP).
CAR-T Cell-Mediated Cytotoxicity
Antibody-Dependent Cell-Mediated Cytotoxicity (ADCC)
T Cell-Dependent Cell-Mediated Cytotoxicity (TDCC)
Antibody-Dependent Cellular Phagocytosis



![Dose-response curve showing the percentage of antibody-dependent cellular phagocytosis (ADCP) as a function of trastuzumab concentration (log₁₀[g/ml]). The curve demonstrates a sigmoidal increase in ADCP, with maximal activity plateauing at higher concentrations.](/-/media/images/resources/figures/19500-19599/19503ma.svg?rev=59862dbe7eed41ae87a775b15ea3eaaa)
CAR-T activity demonstrated using HiBiT Target Cells. T cells transduced with CAR-19 or a GFP control lentivirus were combined with HiBiT Target Cells (Ramos) at different effector:target (E:T) ratios and incubated for 24 hours.
TCK activity measured using the PBMC ADCC Bioassay. PBMC-mediated killing of Raji (HiBiT) Target Cells by anticancer therapeutic mAb, rituximab.
The TDCC Assay measures target cell-specific killing. Thaw and Use CD8+ T Cells (TDCC Qualified) were combined with HiBiT-expressing Target Cells and appropriate Bispecific T Cell Engagers (BiTEs.)
The Macrophage ADCP Bioassay quantifies phagocytic killing of target cells. Thaw and Use, ADCP-qualified human macrophage were combined with HiBiT Target Cells and trastuzumab to measure ADCP activity.
All HiBiT TCK Assays use one of our Bio-Glo-NB™ Luciferase detection reagents.
ADCC, TDCC and CAR-T applications:
Bio-Glo-NB™ TCK Luciferase Assay System
ADCP applications:
Bio-Glo-NB™ Lytic Luciferase Assay System
Webinars
Tools for Biologics
Learn more about our reagents, tools, and custom services for characterizing biologic drug potency.
Resetting Expectations for CAR-T Potency Assessments
Learn how HiBiT TCK Bioassays can be used for determining CAR-T potency.
Innovative Solutions for ADCC and ADCP in Cell Therapies
Experts share how our technologies streamline ADCC and ADCP measurements.
Featured Resources

Automated Nucleic Acid Extraction for Cell and Gene Therapy
Learn how to automate DNA and RNA extraction from a variety of cells and tissues using Maxwell® Instruments.
Video: Target Cell Killing Bioassays
Learn about a solution for measuring CAR-T cell potency in this video interview with Promega scientist Dr. Julia Gilden.