Trouble-Free SDS-PAGE Analysis of Proteins Synthesized in TNT® Cell-Free Expression Systems
Natalie Betz
Promega Corporation
Publication Date: 2004
Abstract
We examined the effects of nonoptimal SDS polyacrylamide gel electrophoresis (SDS-PAGE) loading and running conditions on the results of cell-free protein expression reactions using the TnT® T7 Quick for PCR DNA System. Nonoptimal conditions can result in anomalous or uninterpretable results.
In vitro transcription/translation is a simple and fast method to synthesize small amounts of a protein or to confirm the presence of an open reading frame. The newly synthesized protein is often analyzed by SDS-polyacrylamide gel electrophoresis (SDS-PAGE) to estimate the size of the protein produced. We have found that a number of factors, including the denaturation temperature and the volume of TnT® reaction loaded per well, can affect SDS-PAGE and cause anomalous or uninterpretable results. In this article, we examine the effect of these factors and show examples of problems observed under nonoptimal conditions.
Methods
In vitro transcription/translation reactions were performed with the TnT® T7 Quick for PCR DNA System (Cat.# L5540) as directed in the TnT® T7 Quick for PCR DNA System Technical Manual #TM235 (1). Reactions contained 4µl of 35S-methionine (Amersham Cat.# AG1094) and 500ng (5µl) of the T7 Control DNA (Cat.# L4821), linearized with Eco47III or uncut (supercoiled). The T7 Control DNA encodes the 61kDa firefly luciferase protein. Negative control reactions with no DNA or 500ng of supercoiled pSP72 (Cat.# P2191) were also set up. Reactions were incubated at 30°C for 90 minutes. Aliquots of 1–10µl of each reaction were mixed with 5µl of 4X NuPAGE® LDS Sample Buffer and 2µl of NuPAGE® Sample Reducing Agent and heat denatured at either 70°C for 10 minutes, 85°C for 5 minutes or 95–100°C for 2 minutes. Samples were separated on a 4–12% Bis-Tris NuPAGE® polyacrylamide gel and run in NuPAGE® MES SDS Running Buffer in the presence of NuPAGE® Antioxidant. Once the dye front had reached the bottom of the gel, the gel was fixed for 30–60 minutes in 50% methanol/10% acetic acid, then soaked for 5 minutes in 7% methanol/7% acetic acid/1% glycerol and dried for 1 hour at 70°C under vacuum on a gel dryer. The dried gels were exposed to autoradiograph film at room temperature for 3–16 hours.
Results
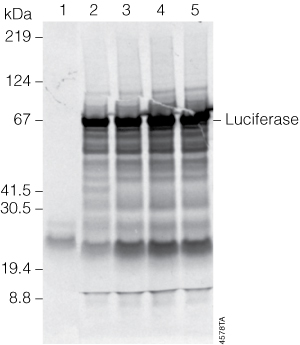
To examine the effect of denaturation conditions, 2µl of a TnT® reaction was denatured at 70°C for 10 minutes, 85°C for 5 minutes or 95–100°C for 2 minutes prior to SDS-PAGE. The results are shown in Figure 1. Very few background bands were seen in the negative control reaction (no DNA template, lane 1). For this control template and amount of reaction loaded per well (2µl), all three denaturation conditions appeared comparable. However, feedback from other users of TnT® Systems suggests the results of denaturation are protein-dependent and that some proteins can migrate as higher molecular weight proteins, presumably protein aggregates, when denatured at higher temperatures.
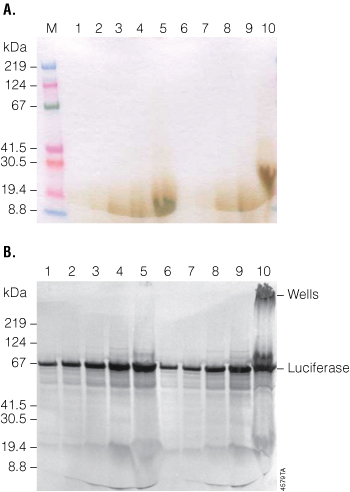
To investigate the effect of loading increasing volumes of a TnT® reaction onto a gel, we denatured increasing amounts of the reaction with the T7 Control DNA (1, 2, 4, 6 or 10µl). Denaturation was performed at 70°C for 10 minutes or 95–100°C for 2 minutes. The samples were separated by SDS-PAGE, and the dried gels were photographed. Figure 2, Panel A, illustrates how unevenly the protein migrated in lanes containing 6µl or 10μl of the TnT® reaction. The lane containing 10µl of the TnT® reaction was particularly distorted when denatured at the higher temperature. The corresponding autoradiograph (Figure 2, Panel B) also shows that the lanes containing 6–10µl of the TnT® reaction are quite overloaded, especially the 10µl lane. In addition, denaturation of samples containing more protein (i.e., the 10µl sample in lane 10) showed high-molecular weight protein retained in the well of the gel (lane 10), while this did not occur when the denaturation was performed at 70°C for 10 minutes (lane 5). We do not recommend loading more than 5µl of a TnT® reaction in a single lane of a polyacrylamide gel, and when loading larger volumes, protein denaturation should be performed at 70°C for 10 minutes for optimal sample migration.
Summary
We have determined that the way in which TnT® reactions are prepared prior to SDS-PAGE can affect the results and can complicate interpretation of the data. The rabbit reticulocyte lysate used in the TnT® Systems has a high protein concentration (100–150µg/µl), which increases the risk of protein aggregation upon heat denaturation and overloading of an SDS polyacrylamide gel. To avoid distortions associated with these phenomena, we recommend limiting the volume of TnT® reaction loaded per lane to no more than 5μl. We also recommend denaturing proteins at 70°C for 10 minutes rather than the standard 95–100°C for 2–3 minutes, especially when volumes larger than the recommended 1–2µl are loaded on the gel. For more information on optimizing your TnT®reactions, see Optimize Your TnT® Reticulocyte Lysate Systems Reactions.
Learn more about the TnT® T7 Quick for PCR DNA