Compatibility of Pierce BCA Protein Assay with Promega Lysis Buffers and Lytic Reagents
Natalie Betz, Promega Corporation
Publication Date: 2003
Abstract
Introduction
Researchers often need to determine the protein concentration of cell lysates used in reporter assays to determine specific activity. Lysis buffers can contain components, such as detergents or reducing agents, that can interfere with protein quantitation assays. The Pierce BCA Protein Assay is one of the most commonly used protein quantitation assays, so we have determined the compatibility of this assay with each of the lysis buffers or the reagents containing a lytic agent used in Promega reporter assays. All of the lysis buffers and reagents were tested at various dilutions, ranging from 1X to 0.001X. The lysis buffers/reagents were diluted to determine the concentration at which they become compatible with the BCA Protein Assay. For some lysis buffers/reagents, the compatible dilution is not practical for actual protein concentration determination, and such reagents will require the use of detergent-compatible and reducing agent-compatible protein assays, of which there are 3 commercially available (Table 1).
Methods and Results
BSA standard curves (between 0–1,000µg/ml) were prepared in either water (control) or various dilutions of the following lysis buffers or resuspended reagents: Reporter Lysis Buffer (RLB; Cat.# E3971), Cell Culture Lysis Reagent (CCLR; Cat.# E1531), Passive Lysis Buffer (PLB; Cat.# E1941), Glo Lysis Buffer (GLB; Cat.# E2661), Renilla Luciferase Assay Lysis Buffer (RLALB; Cat.# E2810, E2820), Steady-Glo® Reagent (Cat.# E2510, E2520, E2550), Bright-Glo™ Reagent (Cat.# E2610, E2620, E2650), Dual-Glo® Reagent (Cat.# E2920, E2940, E2980), CellTiter-Glo® Reagent (Cat.# G7570, G7571, G7572, G7573), Apo-ONE® Reagent (Cat.# G7790, G7791, G7792), and the lysis buffer recommended in the Emax® ImmunoAssay Systems (referred to as Emax® lysis buffer).
An aliquot of BSA stocks (2µl) was added to 23µl of each lysis buffer/reagent. A 25µl sample volume is recommended for the microplate procedure of the BCA Protein Assay. The BCA Protein Assay was performed following the manufacturer's protocol. The absorbance at 562nm was read in a SpectraMAX® 250 plate reader. Each assay was performed in duplicate, and the results are presented as the average of duplicates. In general, the duplicates varied by <5%.
Figures 1–11 illustrate the BSA standard curves generated in each of the lysis buffers or reconstituted reagents. The BSA standard curve generated in water is the reference (+ control) in each graph. Table 1 summarizes all of the results with the recommended dilution for optimal compatibility with the BCA Protein Assay. Preliminary results demonstrated that the potential light output from the Bright-Glo™/Steady-Glo®/Dual-Glo®/CellTiter-Glo® Reagents in the presence of their respective enzymatic substrates did not interfere with or enhance background readings at 562nm (data not shown).
Figures and Tables
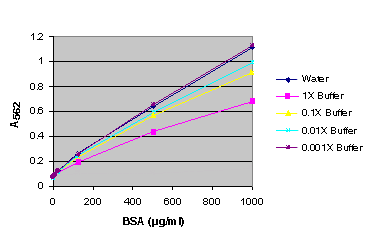
Figure 1. BSA standard curve generated in either water or various dilutions of Reporter Lysis Buffer (RLB).
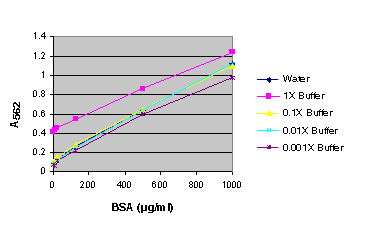
Figure 2. BSA standard curve generated in either water or various dilutions of Cell Culture Lysis Reagent (CCLR). BSA standard curve using Cell Culture Lysis Reagent (CCLR).
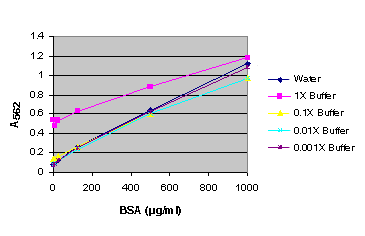
Figure 3. BSA standard curve generated in either water or various dilutions of Passive Lysis Buffer (PLB). BSA standard curve using Passive Lysis Buffer (PLB).
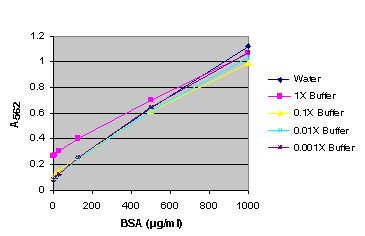
Figure 4. BSA standard curve generated in either water or various dilutions of Glo Lysis Buffer (GLB). BSA standard curve using Glo Lysis Buffer (GLB).
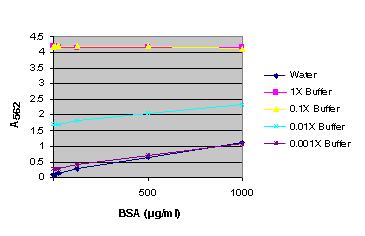
Figure 5. BSA standard curve generated in either water or various dilutions of Renilla Luciferase Assay Lysis Buffer (RLALB). BSA standard curve using Renilla Luciferase Assay Lysis Buffer (RLALB).
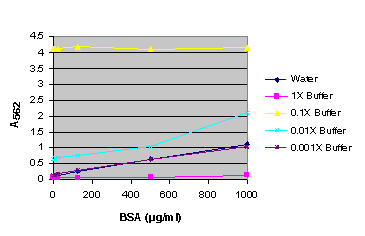
Figure 6. BSA standard curve generated in either water or various dilutions of Steady-Glo® Reagent.
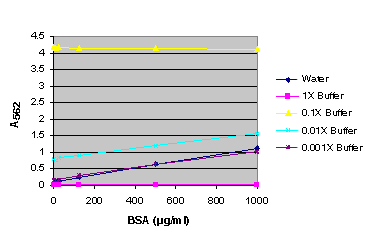
Figure 7. BSA standard curve generated in either water or various dilutions of Bright-Glo™ Reagent.
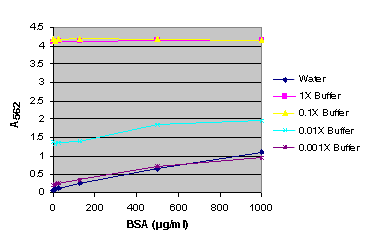
Figure 8. BSA standard curve generated in either water or various dilutions of Dual-Glo® Reagent.
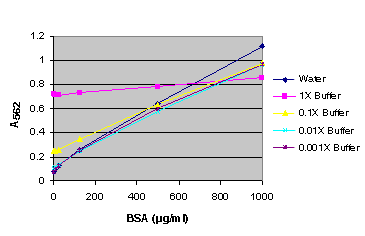
Figure 9. BSA standard curve generated in either water or various dilutions of CellTiter-Glo® Reagent.
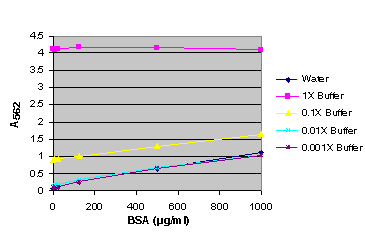
Figure 10. BSA standard curve generated in either water or various dilutions of Apo-ONE® Reagent.
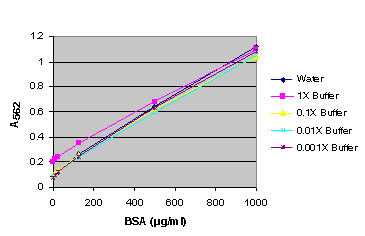
Figure 11. BSA standard curve generated in either water or various dilutions of Emax® lysis buffer.
Table 1.
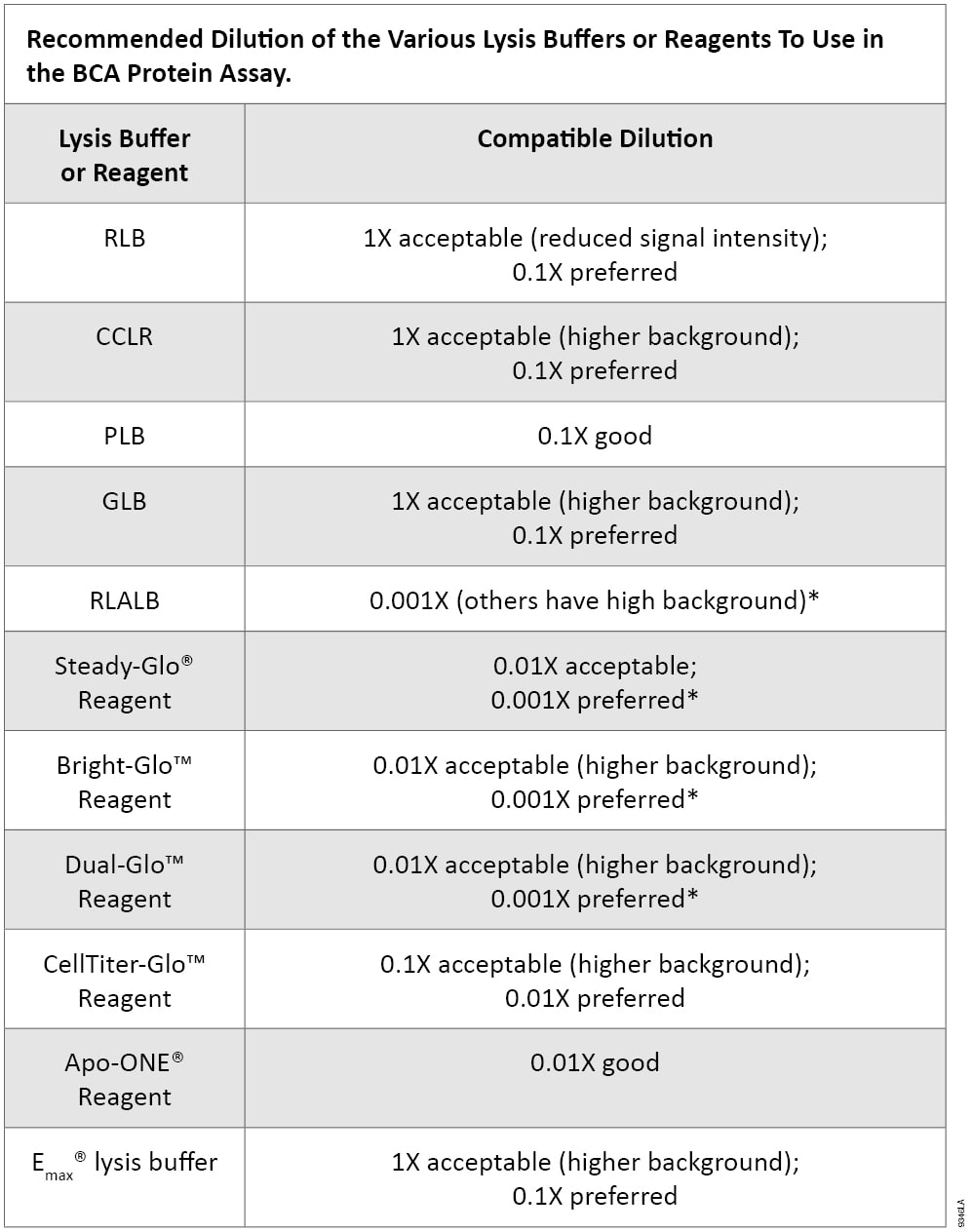
*Use a detergent- and reducing agent-compatible protein assay, such as:
- Geno Technology NI™ Protein Assay (Cat.# 786-005)
- Pierce BCA Protein Assay in conjunction with the Compat-Able™ Protein Assay Preparation Reagent Set (Cat.# 23225 or 23227 and Cat.# 23215)
- Bio-Rad RC DC™ Protein Assay (Cat.# 500-0121 or 500-0122)
Note: When performing a protein assay using any of the suggested protein assay systems, an equal concentration of lysis buffer or reagent must be included in the standard curve for optimal accuracy.
Learn more about Promega Reporter Assays and their Buffers