Differentiating Changes in Glutathione Levels from Cytotoxic Events Using Multiplexed Assays
Alisha Truman and Brad Hook
Promega Corporation
Publication Date: October 2013
Abstract
Measuring changes in glutathione levels as a single endpoint may not be sufficiently informative to differentiate a decrease in GSH from a cytotoxic event. Cytotoxicity assays are often set up in parallel samples. The cytotoxic and oxidative effects of test compounds on cultured cells can be determined using the CellTox™ Green Cytotoxicity Assay and the GSH/GSSG-Glo™ Assay.
Here we demonstrate that both assays can be multiplexed in a single well. Using HEK-293 adherent cells and K-562 suspension cells treated with varying concentrations of menadione or l-Buthionine Sulfoximine (BSO), two compounds known to affect total glutathione (GSH + GSSG) levels, we assayed the cells in separate assays and in a multiplex assay. We show that the addition of CellTox™ Green Dye to the cells does not have a significant effect on the subsequent luminescent signal generated from the GSH/GSSG-Glo™ Assay.
Introduction
Glutathione is an antioxidant found in eukaryotic cells, most often in the reduced form (GSH). A small portion of cellular glutathione is in the oxidized state (GSSG), which takes on the form of a dimer connected by a disulfide bond between the cysteines of two glutathione molecules. GSSG is thus an excellent indicator of oxidative stress in cells.
The GSH/GSSG-Glo™ Assay is a luminescence-based system used to detect and quantify glutathione in both the oxidized and reduced states. The stable luminescence is produced by the conversion of Luciferin-NT, a GSH probe, to luciferin by a glutathione S-transferase enzyme. This conversion to luciferin is coupled to a firefly luciferase reaction that produces a luminescent signal proportional to the amount of GSH present. These measurements allow the calculation of the ratio of GSH/GSSG, or the ratio of reduced to oxidized glutathione, in order to determine the oxidative stress present in cultured mammalian cells in response to a compound.
To quantify glutathione in both the oxidized and reduced forms, the GSH/GSSG-Glo™ Assay uses two reaction configurations to independently quantify GSH and GSSG (Figure 1).

Figure 1. GSH/GSSG-Glo™ Assay Overview. GSH-dependent conversion of a GSH probe, Luciferin-NT, to luciferin by a glutathione S-transferase enzyme is coupled to a firefly luciferase reaction.
The CellTox™ Green Cytotoxicity Assay is a fluorescent, dye-based assay used to determine the cytotoxic effect of experimental compounds on cultured cells. The dye cannot pass through viable cell membranes and thus only binds DNA via the compromised membranes of dead cells (Figure 2). The dye is essentially non-toxic and once bound to DNA produces a stable fluorescent signal for up to 72 hours. This fluorescent signal is proportional to cell cytotoxicity.
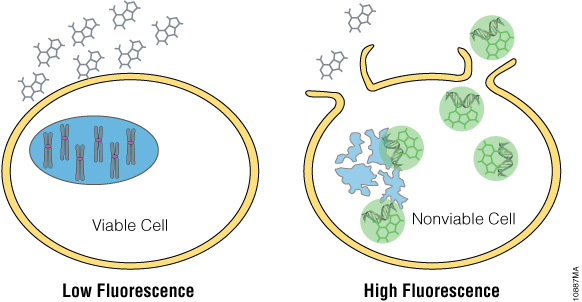
Figure 2. CellTox™ Green Cytotoxicity Assay. CellTox™ Green Dye binds to DNA of cells with impaired membrane integrity and exhibits increased fluorescence.
In this report we demonstrate the efficacy of multiplexing the CellTox™ Green Cytotoxicity Assay with the GSH/GSSG-Glo™ Assay to determine both the cytotoxic and oxidative effects of compounds on cultured cells in a single assay plate. Cultured mammalian cells were treated with menadione or BSO: menadione increases oxidation of glutathione, while BSO inhibits synthesis of cellular GSH. Because the CellTox™ Green Dye can be added in a “no-step” format by adding the dye directly to cells at plating, the CellTox™ Green Cytotoxicity Assay can be performed prior to the GSH/GSSG-Glo™ Assay without changes to either protocol. The experiment was performed in the presence and absence of CellTox™ Green Dye to determine if the fluorescent dye had an effect on the subsequent luminescent signal of the GSH/GSSG-Glo™ Assay.
Materials and Methods
- GSH/GSSG-Glo™ Assay (Cat.# V6611)
- CellTox™ Green Cytotoxicity Assay (Cat.# G8741)
- menadione (Sigma, Cat.# M5625)
- l-Buthionine Sulfoximine (Sigma, Cat.# B2515)
- digitonin (Sigma, Cat.# D141)
- HEK-293 cells
- K-562 cells
Note: Cell lines were authenticated using STR genotyping (GenePrint® 10 System, Cat.# B9510).
Ten thousand cells/well were plated in white 96-well plates (Table 1). Cells were plated in rows B–G and columns 2–10, with 100µl DMEM or Hanks Balanced Salt Solution (HBSS) plated in remaining wells. For each assay, cells were plated in duplicate, with one plate containing CellTox™ Green Dye added according to the Express, No-Step Addition at Seeding protocol described in the CellTox™ Green Cytotoxicity Assay Technical Manual #TM375 (10µl of CellTox™ Green Dye per 5ml of cells). For each treatment, one column contained 30µg/ml of digitonin as a positive control for cytotoxicity. Vehicle control and no-cell control wells also were included.
| Table 1. Assay specifications for multiplexing the CellTox™ Green Assay with the GSH/GSSG-Glo™ Assay. HEK-293 or K-562 cells were treated with menadione or BSO. The cells were assayed for cytotoxicity with the CellTox™ Green Cytotoxicity Assay and for oxidative stress with the GSH/GSSG-Glo™ Assay. | |||
| Assay Step |
Adherent Cells - Menadione |
Adherent Cells - BSO |
Suspension Cells - Menadione |
| Cell Plating (Cells plated in duplicate, with 10µl of CellTox™ Green Dye per 5ml of cells added to one set of duplicate wells) | HEK-293 adherent cells in 100µl DMEM | HEK-293 adherent cells in 50µl DMEM | K-562 suspension cells in 20µl HBSS |
| Incubation at 37° C with 5% CO2 | 24 hours | 4 hours | None |
| Compound Dosing | 10µl menadione (3.125–25µM) |
50µl BSO (15.6µM–500µM) |
5µl menadione (3.125–25µM) |
| Incubation at 37°C with 5% CO2 | 1 hour | 18 hours | 1 hour |
| CellTox™ Green Fluorescence Read | Plate containing CellTox™ Green Dye read at 490nmex/510–550nmem | ||
| GSH/GSSG-Glo™ Assay (Performed as indicated in the GSH/GSSG-Glo™Assay Technical Manual #TM344) | Medium aspirated prior to assay | Assay performed in medium | |
| GSH/GSSG-Glo™ Luminescence Read |
All plates analyzed as indicated in GSH/GSSG-Glo™ Assay Technical Manual #TM344 | ||
Results
We used the GSH/GSSG-Glo™ Assay to monitor GSH and GSSG levels and observe the oxidative stress in adherent HEK-293 cells and suspension K-562 cells dosed with menadione (Figures 3 and 4). The cells also were assayed for the cytotoxicity effects of menadione using the CellTox™ Green Cytotoxicity Assay. Cells dosed with a serial dilution of menadione showed no cytotoxicity at any of the concentrations when compared to the vehicle control. This indicates that the compound did not kill the cells (Figure 3, Panel A, and Figure 4, Panel A). By comparison, the addition of digitonin, a detergent used as a positive control, resulted in a large increase in the cytotoxic signal.
To determine oxidative stress on the cells dosed with menadione, we monitored GSSG levels (Figure 3, Panel B, and Figure 4, Panel B). The oxidized glutathione levels increased with increasing menadione concentrations, indicating oxidase stress. At 25µM of menadione, there was a 13.3-fold increase for HEK-293 cells and 11.5-fold increase for K-562 cells in the amount of oxidized glutathione compared to the control. Total glutathione levels slightly decreased as the concentration of menadione increased (Figure 3, Panel C, and Figure 4, Panel C).
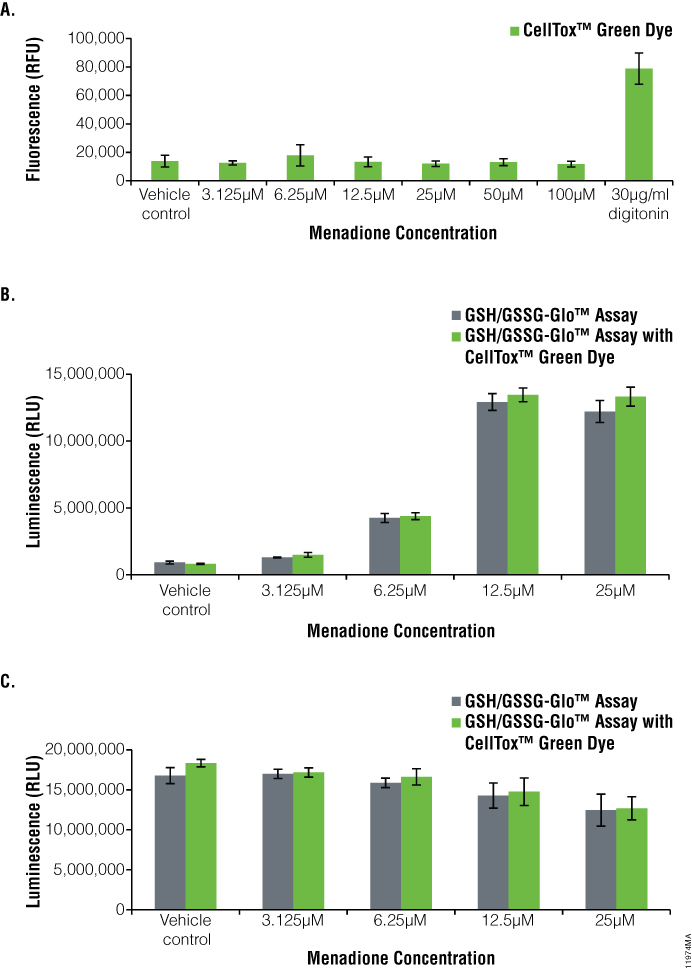
Figure 3. Menadione treatment of HEK-293 cells.The GSH/GSSG-Glo™ Assay was performed on HEK-293 cells after treatment for one hour with menadione in the presence and absence of CellTox™ Green Dye. Panel A. CellTox™ Green Cytotoxicity Assay performed prior to aspiration of medium for GSH/GSSG-Glo™ Assay. Panel B. Oxidized glutathione (GSSG) values. Panel C. Total glutathione values. Gray bars indicate GSH/GSSG-Glo™ Assay only, whereas green bars indicate GSH/GSSG-Glo™ Assay multiplexed with the CellTox™ Green Cytotoxicity Assay.
There was a 1.4-fold decrease for HEK-293 cells and a 1.1-fold decrease for K-562 cells in total glutathione levels at 25µM of menadione compared to the vehicle control. With CellTox™ Green Dye present, there was only a slight change in oxidized glutathione or total glutathione levels, indicating no adverse effect of multiplexing these two assays (compare green and gray bars, Figure 3, Panels B and C, Figure 4, Panel B and C).
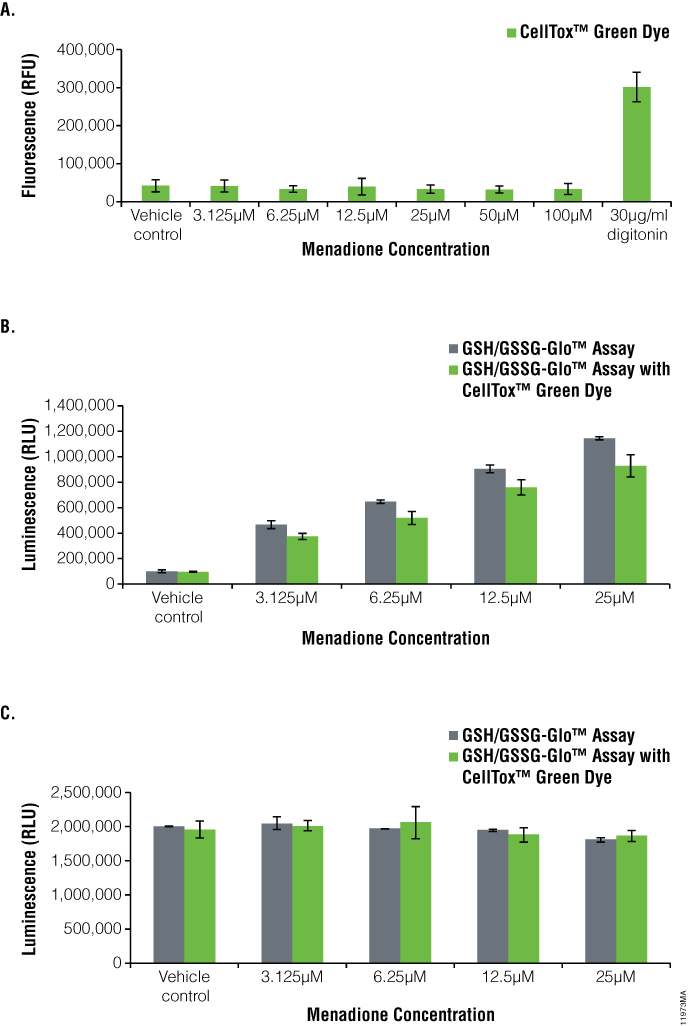
Figure 4. Menadione treatment of K-562 cells.The GSH/GSSG-Glo™ Assay was performed on K-562 suspension cells after treatment for one hour with menadione in the presence and absence of CellTox™ Green Dye. Panel A. CellTox™ Green Cytotoxicity Assay. Panel B. Oxidized glutathione (GSSG) values. Panel C. Total glutathione values. For B/C: Gray bars indicate GSH/GSSG-Glo™ Assay only, whereas green bars indicate GSH/GSSG-Glo™ Assay multiplexed with the CellTox™ Green Cytotoxicity Assay.
To observe GSH changes on adherent HEK-293 cells dosed with l-Buthionine Sulfoximine (BSO), we used the GSH/GSSG-Glo™ Assay to monitor total GSH levels (Figure 5). BSO is a known inhibitor of the synthesis of cellular GSH. Cells also were assayed for cytotoxicity effects of BSO using the CellTox™ Green Cytotoxicity Assay. Cells dosed with a serial dilution of BSO showed no cytotoxicity compared to the vehicle control, indicating that the compound did not kill the cells (Figure 5, Panel A). Adding the positive control, digitonin, resulted in a large increase in cytotoxic signal.
We used the GSH/GSSG-Glo™ Assay to monitor GSH changes in the cells dosed with BSO (Figure 5, Panel B). As the BSO concentrations increased, total glutathione levels decreased as compared to the vehicle control. At a concentration of 25µM BSO, there was a 12.3-fold decrease in the amount of total glutathione compared to the control. BSO did not cause oxidation of glutathione (data not shown). With CellTox™ Green Dye present, there was no significant difference in total glutathione levels detected, indicating no adverse effect of multiplexing these two assays (compare green and gray bars, Figure 5, Panel B).
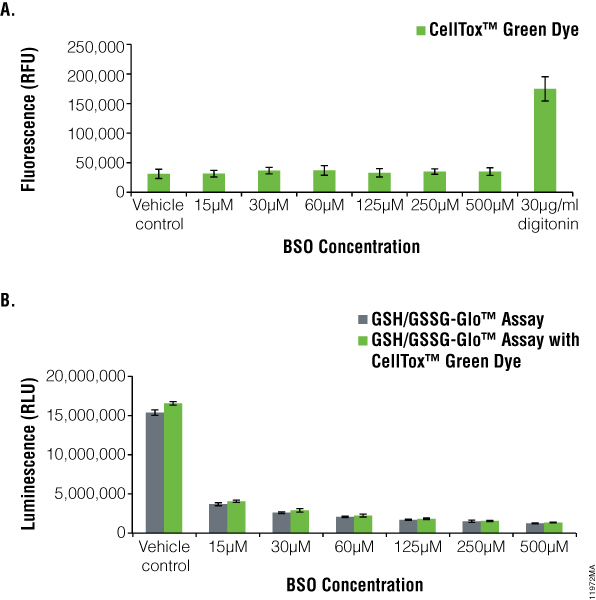
Figure 5. l-Buthionine Sulfoximine (BSO) treatment of HEK293 cells.The GSH/GSSG-Glo™ Assay was performed on HEK-293 cells after treatment for 18 hours BSO in the presence and absence of CellTox™ Green Dye. Panel A. CellTox™ Green Cytotoxicity Assay performed prior to aspiration of medium for GSH/GSSG-Glo™ Assay. Panel B. Total glutathione values (BSO did not cause oxidation of glutathione, data not shown). Gray bars indicate GSH/GSSG-Glo™ Assay only whereas green bars indicate GSH/GSSG-Glo™ Assay multiplexed with the CellTox™ Green Cytotoxicity Assay.
Conclusion
The CellTox™ Green Cytotoxicity Assay can be performed with adherent and suspension cells prior to the GSH/GSSG-Glo™ Assay. The presence of CellTox™ Green Dye does not have a significant effect on the GSH/GSSG-Glo™ Assay luminescent signal. Multiplexing these systems allows measuring oxidative stress and cytotoxicity in the same well with minimal alterations to the protocols.