Comparison of Glucose Uptake Assay Methods
Promega Corporation
Introduction
Glucose is the primary source of energy for many organisms, and the uptake of glucose is a critical process. Glucose is transported across the cell’s membrane and trapped by being phosphorylated. In mammalian cells, this is performed by a family of glucose transporters (GLUT) and a few intracellular hexose kinases. Note that measuring glucose uptake is not the same as measuring glucose consumption. Glucose uptake occurs on a rapid time scale of 10 minutes or less and specifically measures transporter activity, whereas changes in glucose concentration involve a multitude of pathways and typically take several hours.
Why Measure Glucose Uptake?
We can learn several things from measuring glucose uptake. Changes in glucose uptake can reflect overall changes in metabolism, but there are many specific processes as well. In cancer cells, measuring glucose uptake can monitor the overexpression of glucose transporters or identify glucose transporter inhibitors. With fat and muscle cells, changes in GLUT4 translocation upon insulin stimulation can be observed by measuring glucose uptake. In immunologically relevant cells, measuring glucose uptake can be used to follow the transformation of certain cell types from one stage to another.
|
System
|
Model Cells
|
Purpose
|
|---|---|---|
|
Fat and muscle cells |
3T3-L1, 3T3-L1-MBX, L6, C2C12 |
Monitor GLUT4 translocation in response to insulin |
|
Immunologically relevant cells |
T cells and macrophages |
Monitor cell activation |
|
Cancer cells |
Any cancer cell (e.g., HCT116) |
Monitor glucose transporter overexpression, identify glucose transporter inhibitors or measure general changes in metabolism |
Traditional Glucose Uptake Methods
Available glucose uptake assays differ by the substrate for transport. Since glucose itself is shuttled into a variety of pathways after transport, non-metabolizable glucose analogs must be used instead. One type is the fluorescently tagged 2-NBDG (2-(N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino)-2-deoxyglucose). This probe accumulates in the cell and works well for imaging, but because it is much larger than glucose, there is concern that it is not transported in the same way and hence may not be an accurate indicator of glucose transporter activity. Another glucose analog is 2DG (2-deoxyglucose). This molecule gets transported and phosphorylated just like glucose, but since it is not acted upon significantly by other cellular enzymes, it accumulates in the cell as 2DG6P (2-deoxyglucose-6-phosphate).
|
Assay
|
Principle
|
Advantages
|
Disadvantages
|
|---|---|---|---|
|
Radioactivity |
Intracellular accumulation of 3H-2DG6P |
|
|
|
Fluorescence |
Enzymatic detection of 2DG6P |
|
|
|
Absorbance |
Enzymatic detection of 2DG6P |
|
|
|
2-NBDG |
Intracellular accumulation of a fluorescently labelled glucose analog |
|
|
|
Luminescence |
Enzymatic detection of 2DG6P |
|
|
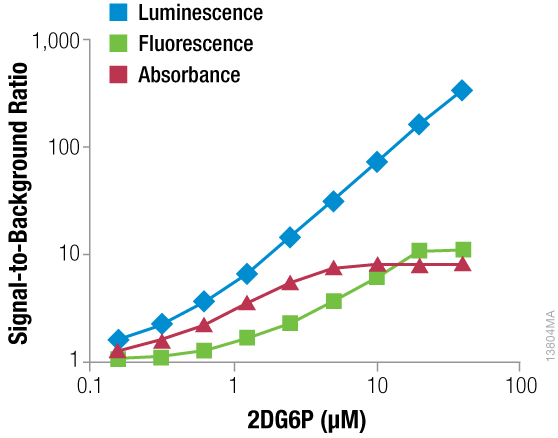
For a long time, the gold standard for measuring glucose uptake has been the radio-labeled analog 3H-2DG and the measurement of accumulated 3H-2DG6P (1). Although the method is sensitive and works well, it requires multiple wash steps. Many researchers do not use this method because they want to avoid the handling and disposal of radioactive material. Commercial non-radioactive methods use non-radiolabeled 2DG and measure the accumulation of 2DG6P through the action of G6PDH (glucose-6-phosphate dehydrogenase) (2). This enzyme oxidizes 2DG6P to form NADPH, which can then be used to generate probes for absorbance or fluorescence assays. Both assays are amenable to multiwell plate formats, but they require multiple processing steps. Due to a cycling detection reaction, the absorbance assay is most sensitive and can detect very low amounts of 2DG6P. However, both assays have small signal windows (i.e., signal above no 2DG6P background) that limit their use.
Glucose Uptake-Glo™ Assay
We have developed a luminescent glucose uptake assay that is based on the same principle as the aforementioned assays: The accumulation of 2DG6P and detection through the enzymatic action of G6PDH (3). The luminescent assay has a number of advantages:
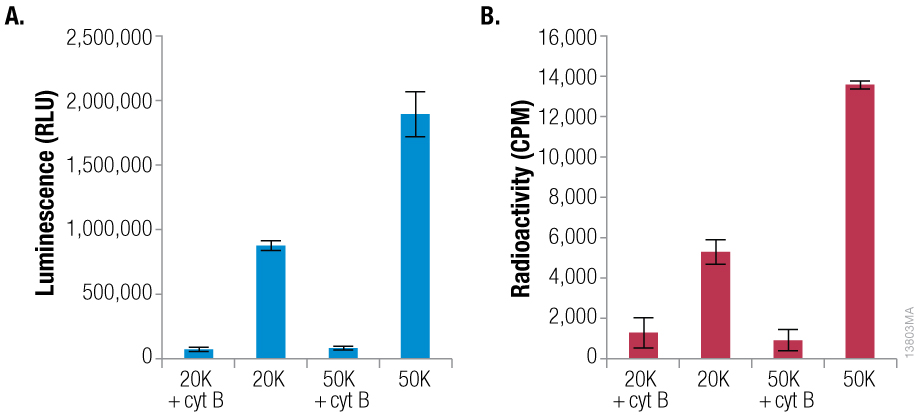
- Similar sensitivity to the radioactive assay but without any wash steps or handling and disposal of radioactive material.
- Larger signal window than the absorbance and fluorescence assays, making it easier to detect small changes and requiring less assay optimization.
- Amenable to high-throughput screening due to fewer false hits and steady glow-type signals.
The luminescent assay can detect glucose uptake from as few as 5,000 cells with a greater than threefold signal-to-background ratio, and the signal remains linear up to at least 50,000 cells. The luminescent assay is the best combination of simplicity and sensitivity available for a non-radioactive multiwell plate method.

References
- Yamamoto, N. et al. (2015) Measurement of glucose uptake in cultured cells. Curr. Protoc. Pharmacol. 71, 12.14.1–26.
- Saito, K. et al. (2011) An enzymatic photometric assay for 2-deoxyglucose uptake in insulin-responsive tissues and 3T3-L1 adipocytes. Anal. Biochem. 412, 9–17.
- Valley, M.P. et al.(2016) A bioluminescent assay for measuring glucose uptake. Anal. Biochem. 505, 43–50.