Lynch Syndrome Detection: The Importance of Determining Status
In some families, cancer occurs more often than expected. A history of an immediate family member (parent, brother, sister) or extended family members with colorectal cancer or polyps could indicate an inherited increased risk for cancer, called Lynch syndrome. Knowing Lynch status can help patients and healthcare professionals make better, more informed decisions before the onset of cancer or before a second cancer has had a chance to develop.

Genetics of Lynch Syndrome
Lynch syndrome, or hereditary nonpolyposis colon cancer, is the most common form of hereditary colorectal cancer and causes roughly 3–5% of all colon cancers(3). Lynch syndrome is caused by autosomal dominant mutations to the major mismatch repair genes MLH1, MSH2, MSH6 or PMS2 as well as the EPCAM gene that inactivates MSH2. In addition to colorectal cancer, Lynch syndrome is associated with several other cancers including endometrial, ovarian and stomach cancer.
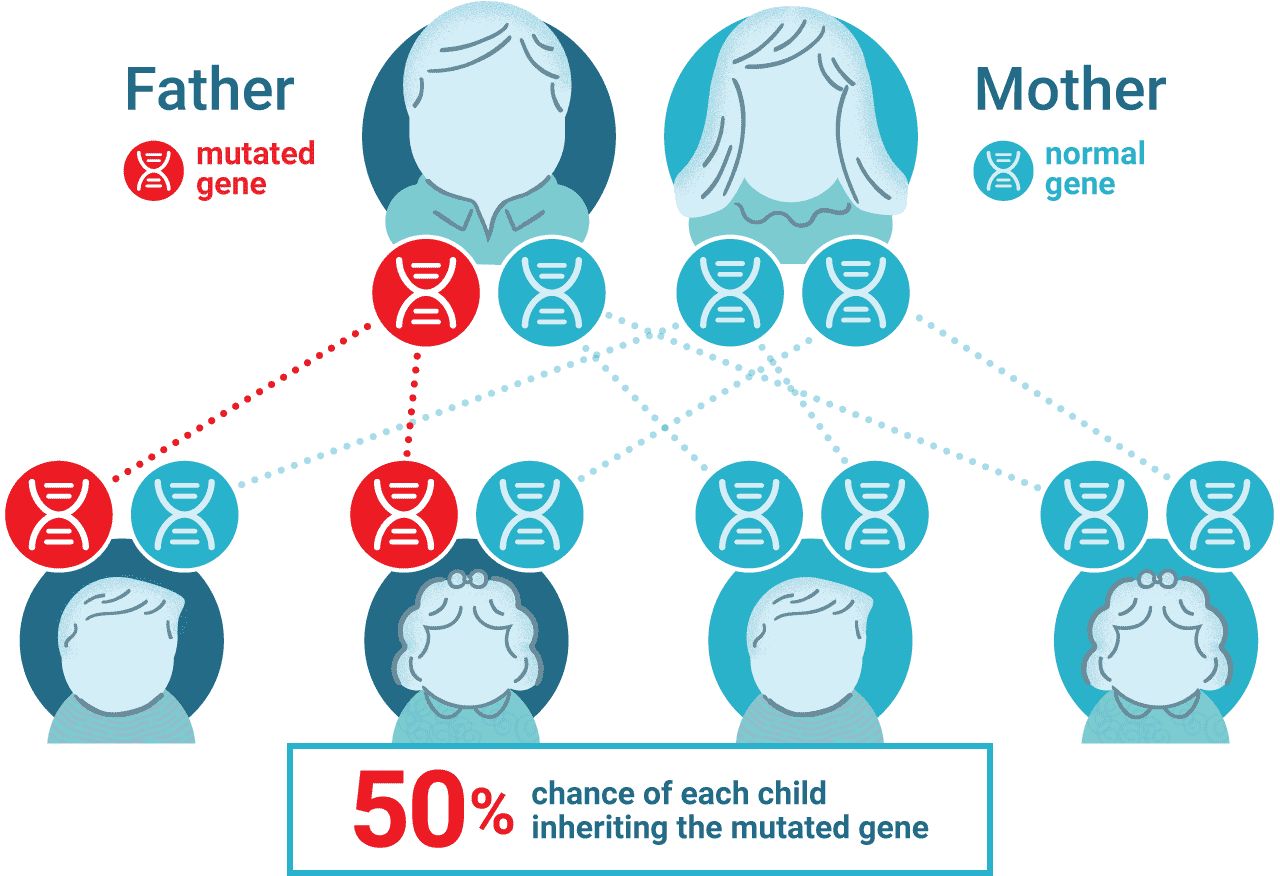
Why is Lynch Status Important?
In addition to colorectal cancer, people with Lynch syndrome have an increased lifetime risk of developing certain other cancers. Because it is an inherited condition, family members might also have the condition and be at risk.
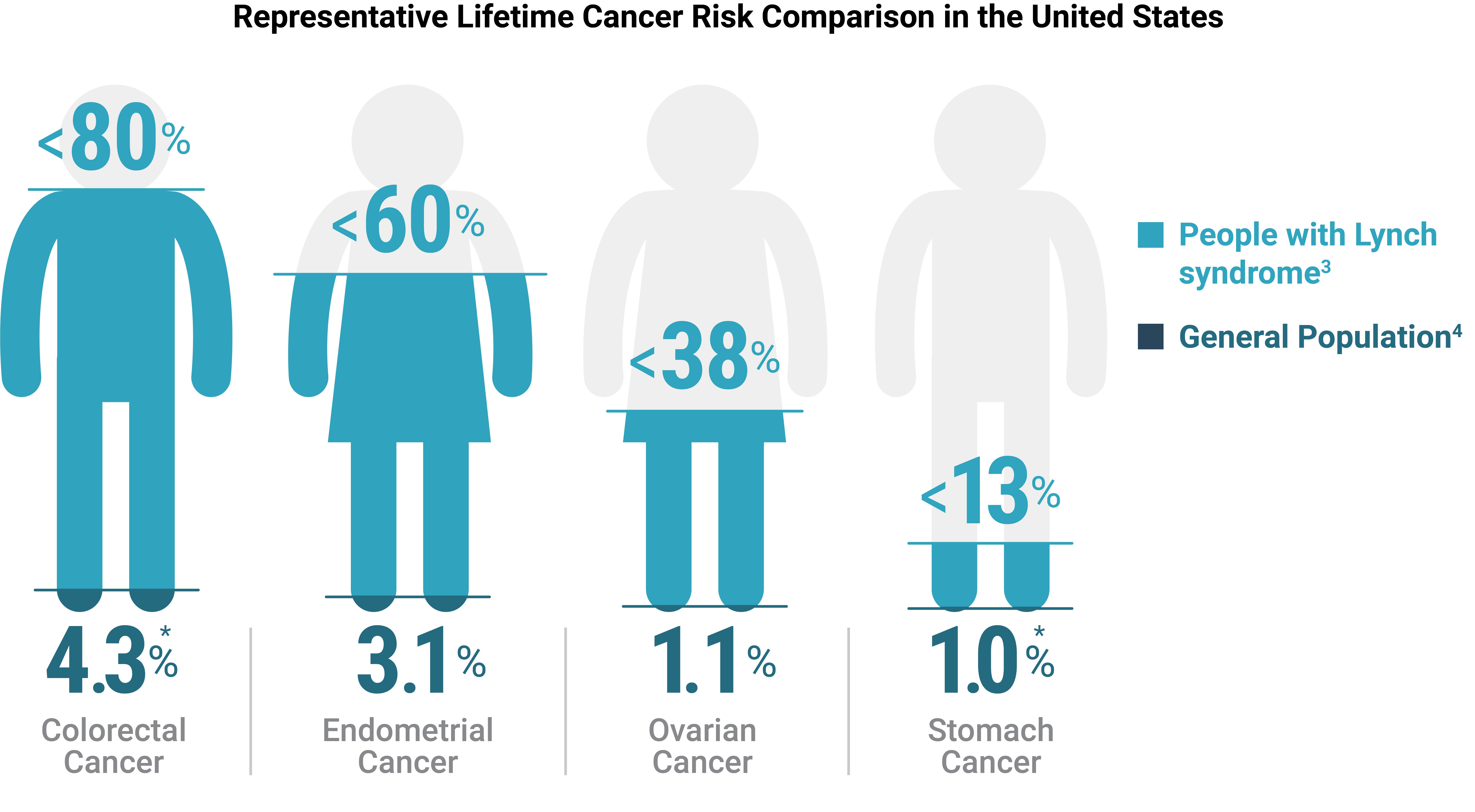
*Value shown for males, lifetime risk for females is 3.9% (colon) and 0.6% (stomach).
How Is Lynch Syndrome Diagnosed?
To determine if Lynch syndrome is likely, simple screening tests can be performed on tumor (cancer) tissue. The two most common tests are microsatellite instability testing (MSI) and immunohistochemistry testing (IHC) for major mismatch repair proteins (MMR). The results of these tests can indicate whether more specific genetic testing should be considered.
Lynch Syndrome and MSI
High microsatellite instability (MSI-H) in tumor tissue is a hallmark of Lynch syndrome. MSI-H status indicates that certain sections of DNA called microsatellites have become unstable because the major mismatch repair proteins that correct errors during DNA replication are not functioning properly. A DNA analysis test is used to determine MSI status. The test is performed on both normal and tumor tissue. If the results for the tumor differ from the normal tissue results, the tumor is considered MSI-H.
Lynch Syndrome and IHC
Immunohistochemistry is a screening test for Lynch syndrome tumors that looks for the proteins expressed by the major mismatch repair (MMR) genes. If the genes are functioning properly, the proteins will be present. Absence of a major mismatch repair protein increases the likelihood that Lynch syndrome is present.
The Life-Saving Power of Lynch Syndrome Detection
With Lynch syndrome, understanding the genetics, recognizing the symptoms, and being informed about screening solutions can empower people to better manage their health. The article “Altering Genetic Destiny: The Life-Saving Power of Lynch Syndrome Detection” offers more information about Lynch syndrome and features a video about this inherited condition that includes an interview with Dr. Heather Tomlinson (Director of Clinical Diagnostics, Promega) discussing how MSI testing can play an important role in identifying Lynch Syndrome.

Dr. Heather Tomlinson
Director of Clinical Diagnostics
Promega Corporation
The Value of Screening Every Tumor for Microsatellite Instability
Universal screening of all tumors and Lynch-associated cancers could help identify patients and families who have an increased risk for colorectal and other cancers. Screening can also identify tumors most likely to respond to immunotherapeutics. Genetic counselor Heather Hampel is an advocate for universal tumor screening for Lynch syndrome.

“…you have tumor tests you can use to identify the patients that are more likely to have it […] And you have surveillance that really works” — Heather Hampel, MS, LGC, The Ohio State University
References:
- Win, A.K. et al. (2016) Prevalence and Penetrance of Major Genes and Polygenes for Colorectal Cancer. Cancer Epidemiol Biomarkers Prev. 26(3), 404–412.
- Cancer.gov. Cancer MoonshotSM Blue Ribbon Panel. Accessed March 16, 2022.
- Cancer.net. Lynch Syndrome. Accessed February 9, 2022.
- Cancer.org. Lifetime Risk of Developing or Dying from Cancer. Accessed January 26, 2022.