Development of the PowerPlex® 21 System
Promega Corporation
Publication Date: 2012
Introduction
As human identification through the use of STR genotyping continues to expand and mature, new typing systems are needed to meet the evolving needs of the human identification community. In Europe, the US and China, STR typing is moving to include more loci to aid discrimination. With these changes, larger multiplexes are required to satisfy locus selection within different regions. In addition to a need for greater discrimination, there is a demand for robust systems to handle more difficult samples in forensic casework as well as a need to process database samples with less upfront preparation. Increased robustness means higher success for forensic casework and faster processing time for offender databasing and paternity testing.
The PowerPlex® 21 System
The PowerPlex® 21 System (Cat.# DC8902) was released in December of 2011 to expand the options available for multiplex STR analysis. This five-color system (Figure 1) includes all materials needed for co-amplification and detection of 21 loci (20 STR loci and Amelogenin), including D1S1656, D2S1338, D3S1358, D5S818, D6S1043, D7S820, D8S1179, D12S391, D13S317, D16S539, D18S51, D19S433, D21S11, Amelogenin, CSF1PO, FGA, Penta D, Penta E, TH01, TPOX and vWA. One primer for each locus is labeled with fluorescein (blue), JOE (green), TMR-ET (yellow) or CXR-ET (red). Detection is performed with the Applied Biosystems 3100, 3130 or 3500 Genetic Analyzer. Amplified samples can be sized with the supplied CC5 Internal Lane Standard 500 (CC5 ILS 500), detected as a fifth dye. Proper color deconvolution is achieved by matrix standardization or spectral calibration using the PowerPlex® 5-Dye Matrix Standards (Cat.# DG4700). Finally, a new control DNA, 2800M, is provided in the system. This DNA is derived from a single-source, human blood sample.
Increased Discrimination
As databases grow in size, the potential for adventitious matches increases. Some early STR databases built with only ten loci have been recognized as insufficient to avoid these unrelated matches once millions of profiles were included in the databases. Another challenge is data sharing between regions that use different STR multiplex products that do not contain the same loci, leading to differences in database composition.
The PowerPlex® 21 System increases discriminatory power and data-sharing possibilities by incorporating informative loci commonly used in the United States, Europe and Asia (Table 1). The PowerPlex® 21 System includes the 13 CODIS core STR loci, two loci commonly used in Europe (D1S1656 and D12S391), the D6S1043 locus common in China, and several additional markers used throughout the world (Amelogenin, Penta D, Penta E, D2S1338 and D19S433).
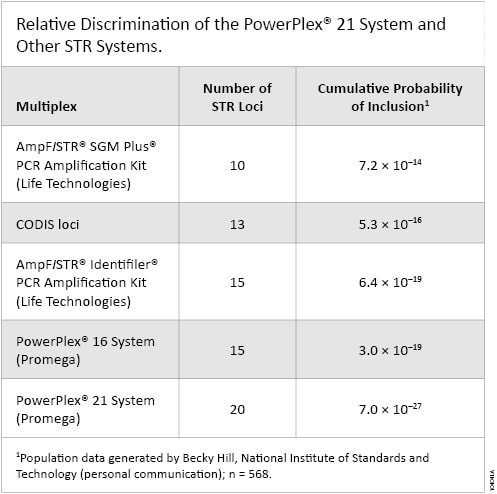 Table 1. Relative Discrimination of the PowerPlex® 21 System and other STR systems.
Table 1. Relative Discrimination of the PowerPlex® 21 System and other STR systems. Unbeatable Inhibitor Tolerance
Forensic evidentiary samples often contain substances that inhibit PCR. Even following a DNA purification step, these inhibitory components can persist, decreasing the likelihood of STR amplification success. In 2009, Promega introduced the PowerPlex® 16 HS System (1) , which demonstrated a significant increase in inhibitor tolerance over previous STR multiplexes. The PowerPlex® 21 System improves on the PowerPlex® 16 HS System performance for even greater inhibitor resistance (Figure 2), resulting in more interpretable data and less need for re-amplification of samples previously deemed "difficult" due to the presence of inhibitors.
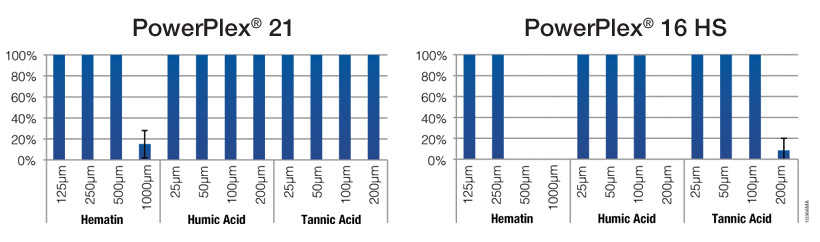 Figure 2. Resistance of the PowerPlex® 21 and 16 HS Systems to PCR inhibitors. Human genomic DNA (500pg; n = 3) was amplified with the PowerPlex® 21 and 16 HS Systems in the presence of common inhibitors such as hematin, humic acid and tannic acid. Percent of alleles called are indicated.
Figure 2. Resistance of the PowerPlex® 21 and 16 HS Systems to PCR inhibitors. Human genomic DNA (500pg; n = 3) was amplified with the PowerPlex® 21 and 16 HS Systems in the presence of common inhibitors such as hematin, humic acid and tannic acid. Percent of alleles called are indicated.Faster Processing of Multiple Sample Types
The advent of direct amplification from FTA® cards has increased demand for faster processing of database, reference and paternity samples. The PowerPlex® 21 System builds on recent advances in Promega STR chemistries, including faster cycling time and direct amplification from FTA® cards. Additionally, the release of this multiplex will be accompanied by new, rapid solutions for swabs and nonFTA cards (SwabSolution™ Kit and PunchSolution™ Kit, respectively). With the new advances, the PowerPlex® 21 System can amplify DNA from a variety of sample types, including difficult casework samples, and is compatible with direct amplification from FTA® card punches as well as pretreated swabs, Bode Buccal Collector™ devices or S&S 903 punches (Figure 3). Finally, rapid cycling technology reduces thermal cycling by as much as two hours. These features combine to reduce sample-to-analysis time for all samples using the PowerPlex® 21 System.
 Figure 3. Amplification of various sample types using the PowerPlex® 21 System.
Figure 3. Amplification of various sample types using the PowerPlex® 21 System. Panel A. Amplification of 500pg of human genomic DNA using the recommended protocol. Amplified products were separated on an Applied Biosystems 3130 Genetic Analyzer (3kV, 5-second injection). Panel B. Direct amplification of one 1.2mm punch from blood on an FTA® card using the recommended protocol. Amplified products were separated on Applied Biosystems 3130 Genetic Analyzer (3kV, 5-second injection). Panel C. A 1.2mm punch from a buccal sample on S&S 903 paper was pretreated with prototype PunchSolution™ reagent, then amplified using the recommended protocol. Amplified products were separated on an Applied Biosystems 3130xl Genetic Analyzer (3kV, 5-second injection). Panel D. A swab from a buccal sample was pretreated with SwabSolution™ reagent, then amplified using the recommended protocol. Amplified products were separated on an Applied Biosystems 3130xl Genetic Analyzer (3kV, 5-second injection).
Conclusion
The PowerPlex® 21 System provides greater discrimination for STR analysis of both casework and database samples than possible with previous multiplexes. Significant labor savings can be realized through simplified protocols for common database and paternity sample types. Increased inhibitor tolerance allows greater success from the most challenging casework samples. Rapid cycling technology ensures that turnaround time for any application is reduced. Finally, in a time of increased data sharing and rapid expansion in the number and size of databases, the PowerPlex® 21 System offers markers used in forensic labs, and found in databases, around the world providing exceptional discrimination to help resolve even more cases.
Related Products
Related Articles
Article References
- Ensenberger, M.G. and Fulmer, P.M. (2009) The PowerPlex® 16 HS System. Profiles in DNA 12(1), 9–11.
How to Cite This Article
Scientific Style and Format, 7th edition, 2006
Ensenberger, M.E. et al. Development of the PowerPlex® 21 System. [Internet] 2012. [cited: year, month, date]. Available from: https://www.promega.com/resources/profiles-in-dna/2012/development-of-the-powerplex-21-system/
American Medical Association, Manual of Style, 10th edition, 2007
Ensenberger, M.E. et al. Development of the PowerPlex® 21 System. Promega Corporation Web site. https://www.promega.com/resources/profiles-in-dna/2012/development-of-the-powerplex-21-system/ Updated 2012. Accessed Month Day, Year.
Contribution of an article to Profiles in DNA does not constitute an endorsement of Promega products.
PowerPlex is a registered trademark of Promega Corporation. PunchSolution and SwabSolution are trademarks of Promega Corporation.
AmpFlSTR, Identifiler and SGM Plus are registered trademarks of Applera Corporation. Bode Buccal DNA Collector is a trademark of the Bode Technology Group, Inc. FTA is a registered trademark of Flinders Technologies, Pty, Ltd., and is licensed to Whatman.