New Control DNA for PowerPlex® Systems
Promega Corporation
Publication Date: 2011
Why should I switch from my current control DNA to 2800M?
If your lab is currently running both autosomal and Y-STR analyses, you are likely using two control DNAs that are specific for each type of analysis. By switching to 2800M Control DNA, you will need only one control DNA for both types of analyses, thereby simplifying your laboratory workflow.
What is the genotype of 2800M Control DNA?
The autosomal STR, Y-STR and Amelogenin genotype for 2800M Control DNA is provided in Table 1. Figure 1 shows the profile for 2800M Control DNA as generated with the PowerPlex® 18D System.
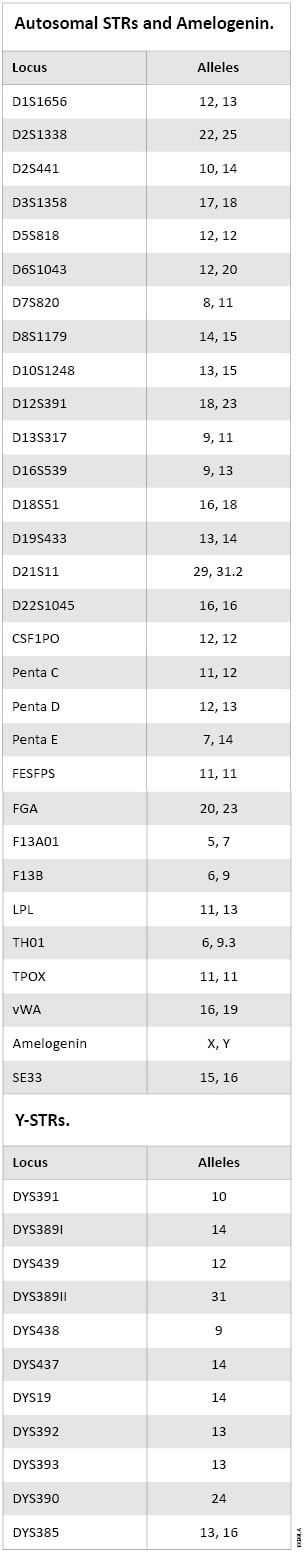 Table 1. Autosomal STR, Amelogenin and Y-STR Genotypes of the New 2800M Control DNA.
Table 1. Autosomal STR, Amelogenin and Y-STR Genotypes of the New 2800M Control DNA. 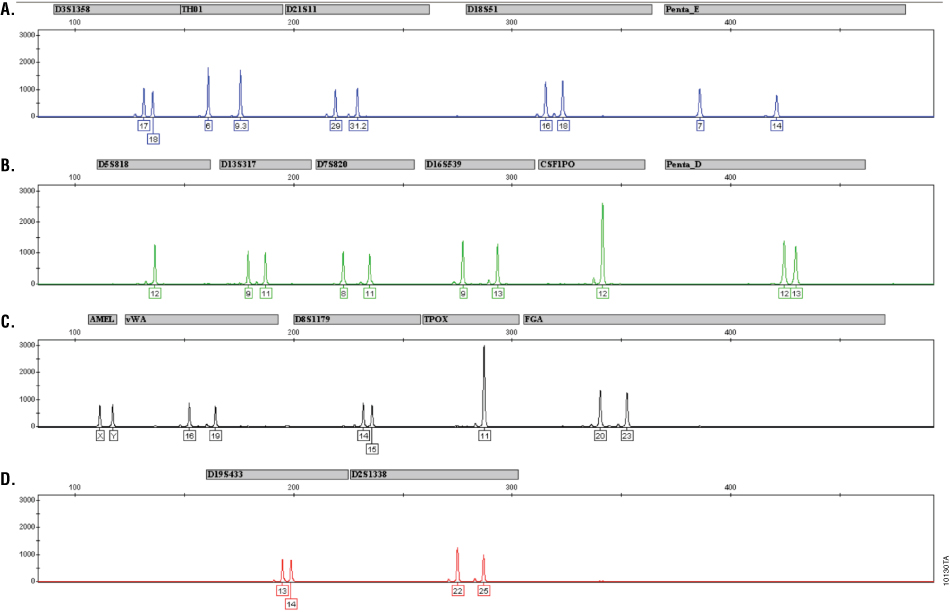 Figure 1. The 2800M Control DNA analyzed using the PowerPlex® 18D System.
Figure 1. The 2800M Control DNA analyzed using the PowerPlex® 18D System.
2800M Control DNA (5ng) was amplified for 27 cycles using the PowerPlex® 18D System. Amplification products were analyzed with an Applied Biosystems 3130xl Genetic Analyzer using a 3kV, 5-second injection. Results were analyzed using GeneMapper® ID software, version 3.2. Panel A. An electropherogram showing the peaks of the fluorescein-labeled loci: D3S1358, TH01, D21S11, D18S51 and Penta E. Panel B. An electropherogram showing the peaks of the JOE-labeled loci: D5S818, D13S317, D7S820, D16S539, CSF1PO and Penta D. Panel C. An electropherogram showing the peaks of the TMR-ET-labeled loci: Amelogenin, vWA, D8S1179, TPOX and FGA. Panel D. An electropherogram showing the peaks of the CXR-ET-labeled loci: D19S433 and D2S1338. The alleles for each locus are indicated.
What is the source of 2800M Control DNA?
The 2800M Control DNA is isolated from the blood of a single human male. It is processed entirely by Promega scientists using proven manufacturing techniques, thus ensuring a consistently high-quality product.
Do I need new GeneMapper® panels and bin sets for use with the 2800M Control DNA?
Yes. Promega has prepared panels and bin sets for both GeneMapper® ID, version 3.2, and GeneMapper® ID-X software for use with the 2800M Control DNA. Select the appropriate PowerPlex® System, GeneMapper® software version and 2800M Control DNA at our request page at: www.promega.com/resources/tools/genemapper-id-software-panels-and-bin-sets/ to download the panels and bin sets.
Do I need to perform validation when I switch to the 2800M Control DNA?
Yes. You should perform a performance check with this new control DNA to confirm the optimal quantity of positive control DNA for your STR amplifications.
Will the 9947A and 9948 control DNAs continue to be available as stand-alone products?
Yes, they will continue to be available as stand-alone products for a limited time. PowerPlex® Systems will be provided with 2800M Control DNA beginning in 2012, except for PowerPlex® 18D, which already includes 2800M Control DNA. K562 DNA (Cat.# DD2011) will continue to be available as a female, human positive control DNA.
Is the 2800M Control DNA appropriate for sensitivity testing?
We recommend that you perform sensitivity testing with samples that are isolated and quantitated using your standard laboratory workflow.
How to Cite This Article
Scientific Style and Format, 7th edition, 2006
James, R. New Control DNA for PowerPlex® Systems. [Internet] 2011. [cited: year, month, date]. Available from: https://www.promega.com/resources/profiles-in-dna/2011/new-control-dna-for-powerplex-systems/
American Medical Association, Manual of Style, 10th edition, 2007
James, R. New Control DNA for PowerPlex® Systems. Promega Corporation Web site. https://www.promega.com/resources/profiles-in-dna/2011/new-control-dna-for-powerplex-systems/ Updated 2011. Accessed Month Day, Year.
Contribution of an article to Profiles in DNA does not constitute an endorsement of Promega products.
PowerPlex is a registered trademark of Promega Corporation.
GeneMapper is a registered trademark of Life Technologies.
Products may be covered by pending or issued patents or may have certain limitations. More information.
All prices and specifications are subject to change without prior notice.
Product claims are subject to change. Please contact Promega Technical Services or access the Promega online catalog for the most up-to-date information on Promega products.