ProTEV Protease for Fusion Protein Processing
Promega Corporation
Publication Date: April 2008
Abstract
The ProTEV Protease is an improved 50kDa version of the Nla protease from Tobacco Etch Virus (TEV), which has been engineered to be more stable than native TEV protease for prolonged enzymatic stability. It is used primarily to cleave affinity tags from fusion proteins during or after affinity purification.
Introduction
Many proteins are expressed as fusion partners with affinity tags, such as glutathione-S-transferase (GST) or maltose binding protein (MBP), to selectively bind the proteins using affinity purification resins. While such resins yield high-purity protein quickly, the large affinity tags are undesirable for some downstream applications. Therefore, most expression vectors are designed with a specific protein cleavage site between the two fusion partners to remove the affinity tag after purification.
A common protease used to remove affinity tags, Factor Xa (FXa; Cat.# V5581), may also cleave the fusion protein because its four-amino acid recognition sequence is quite common. In contrast, ProTEV Protease recognizes a rare amino acid sequence, EXXYXQ, where X is any amino acid and cleavage occurs after the glutamine residue(1)(2). In addition, cleavage sites for TEV protease are found in many common vectors including the thepFN2A and pFN2K (GST) Flexi® Vectors ( Cat.# C8461 and C8471).
Specificity of ProTEV Protease
ProTEV Protease is manufactured to a high standard of purity and is free from nonspecific activity. Overdigestion of a protein panel with ProTEV Protease results in cleavage of only the control protein containing a ProTEV Protease recognition sequence (Figure 1). Since the ProTEV Protease recognition sequence is uncommon, unintentional cleavage of the protein of interest is rare.
 Figure 1. ProTEV Protease has highly specific proteolytic activity.
Figure 1. ProTEV Protease has highly specific proteolytic activity.
Fifteen micrograms of six different proteins were incubated with or without 10 units of ProTEV Protease for 72 hours at 30°C. Digestion of the proteins was analyzed by SDS-PAGE and stained with SimplyBlue™ SafeStain (Invitrogen). Lane M, SeeBlue® Plus2 (Invitrogen); lane 1, ProTEV Protease alone; lanes 2,3, GST-MBP with TEV protease site; lanes 4,5, GST-HaloTag® fusion protein with FXa protease site; lanes 6,7, bovine serum albumin (BSA); lanes 8,9, phosphorylase B (Sigma); lanes 10,11, β-galactosidase (Sigma); lanes 12,13, QuantiLum® Recombinant Luciferase.
Cleavage Conditions for ProTEV Protease
ProTEV Protease is provided with a reaction buffer at pH 7; however, the protease is active over a pH range of 5.5–8.5, allowing it to be used under the conditions most appropriate for the protein of interest. The greatest activity of ProTEV Protease occurs at 30°C, but the protease will cleave fusion proteins over a temperature range of 4–34°C. As with many enzymes, ProTEV Protease cleaves more slowly as the temperature is decreased (Table 1).
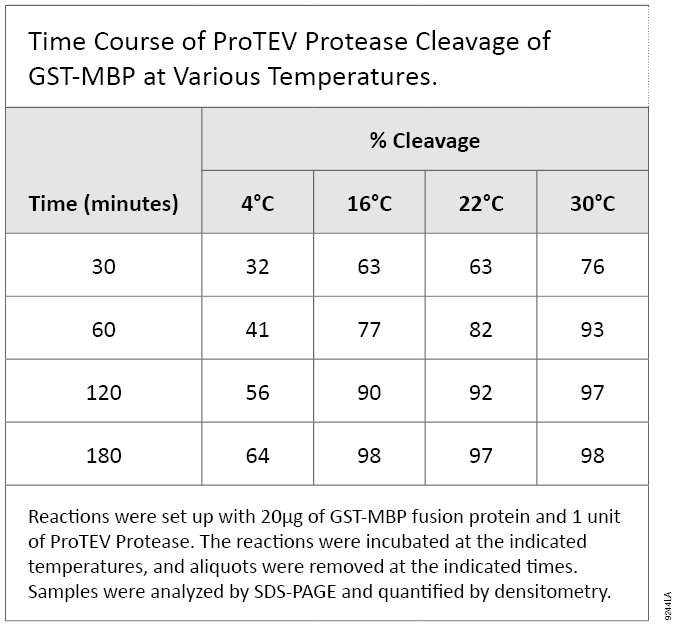 Table 1. Cleavage of GST-MBP at various Temperatures. Reactions were set up with 20µg of GST-MPB fusion protein and 1 unit of ProTEV Protease. The reactions were incubated at the indicated temperatures, and aliquots were removed at the indicated times. Samples were analyzed by SDS-PAGE and quantified by densitometry.
Table 1. Cleavage of GST-MBP at various Temperatures. Reactions were set up with 20µg of GST-MPB fusion protein and 1 unit of ProTEV Protease. The reactions were incubated at the indicated temperatures, and aliquots were removed at the indicated times. Samples were analyzed by SDS-PAGE and quantified by densitometry. For more details on ProTEV Protease, read the full-length Promega Notes article.
Related Products
Related Resources
Related Articles
Article References
- Dougherty, W.G. et al. (1989) Characterization of the catalytic residues of the tobacco etch virus 49-kDa proteinase. Virology 172, 302–10.
- Carrington, J.C. and Dougherty, W.G. (1988) A viral cleavage site cassette: Identification of amino acid sequences required for tobacco etch virus polyprotein processing. Proc. Natl. Acad. Sci. USA 85, 3391–5.
How to Cite This Article
Scientific Style and Format, 7th edition, 2006
Anderson, J., English, J. and Shultz, J. ProTEV Protease for Fusion Protein Processing. [Internet] April 2008. [cited: year, month, date]. Available from: https://www.promega.com/resources/pubhub/enotes/protev-protease-for-fusion-protein-processing/
American Medical Association, Manual of Style, 10th edition, 2007
Anderson, J., English, J. and Shultz, J. ProTEV Protease for Fusion Protein Processing. Promega Corporation Web site. https://www.promega.com/resources/pubhub/enotes/protev-protease-for-fusion-protein-processing/ Updated April 2008. Accessed Month Day, Year.
Products may be covered by pending or issued patents or may have certain limitations on use. Please visit our patent and trademark web page for more information.
Simply Blue is a trademark and SeeBlue is a registered trademark of Invitrogen Corporation.