The Road to Reproducibility
Manuela Grassi, Gretel Celino and Julia Nepper
tpub_218, Oct. 2019
Abstract
One of the great benefits of getting products from a company like Promega is reproducibility: we work hard to make sure that our products perform the same way, every time. How do we do it? I talked to Manuela Grassi, Group Leader for Promega’s Cell Manufacturing, to find out how her team manages to create and QC reproducible quality cell lines.
Manuela told me that her team uses two main strategies to ensure the quality of their cell lines: first, controlling production processing by using environmentally controlled rooms and preapproved manufacturing protocols. Second, they perform a series of quality control tests, such as cell counts, microbial contamination testing and luminescence functional testing, with GloMax® plate readers. The team uses cell-based assay methods that mimic what the customer will do with the cells, i.e., by measuring EC50 and fold induction in response to a specific drug or antibody. Figures 1 and 2 show the results from multiple assay plates measured on three different GloMax® instruments. The data show the consistency of the luminescence response across instruments.
Introduction
Manuela is responsible for leading the team of 11 that manufactures thaw-and-use cell lines for our biologics customers. Before coming to Promega, Manuela worked in vaccine and drug development; ironically, she used the cells that she is now responsible for manufacturing for her cell-based assays. “Cell lines are very complex models to work with,” says Manuela, and “thaw-and-use cells help to control any variables as much as possible” to ensure a consistent response across different days and different plates. Throughput can be another issue with cell-based assays because of the number of replicate samples that are required: by facilitating easy assay setup, thaw-and-use cells allow researchers to get the same data with a lower time investment.
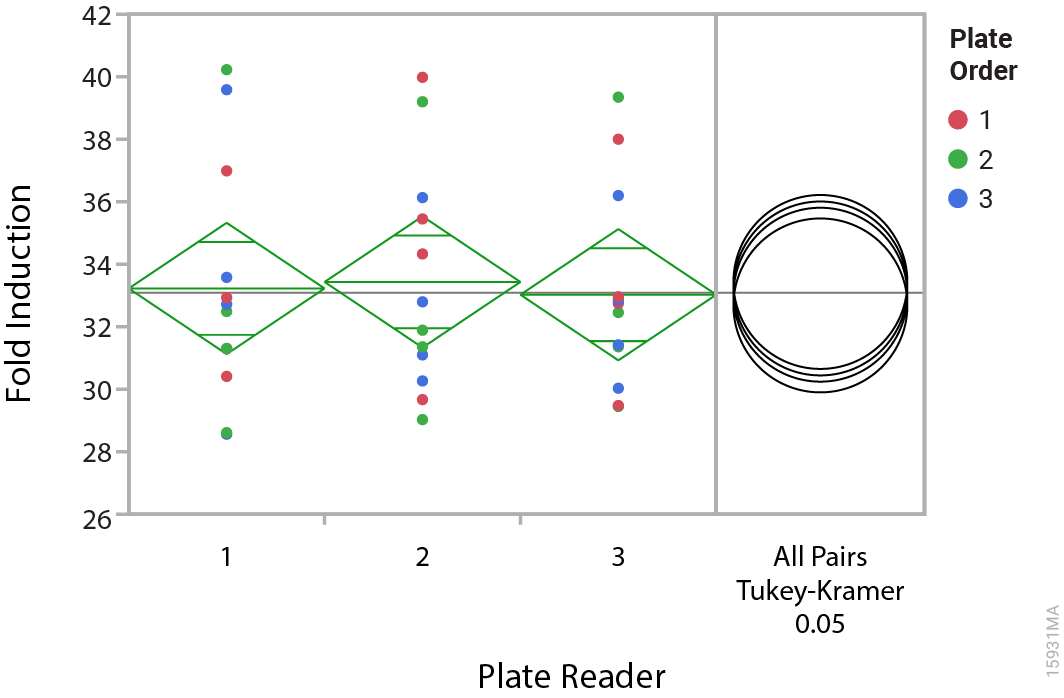
Figure 1. Comparison of fold induction measurements. Multiple ADCC Reporter Bioassays were performed over several days using multiple lots of effector and target (Raji) cell lines and an anti-CD20 antibody. Each bioassay plate was analyzed on three different GloMax® instruments and the results subjected to an analysis of variance (ANOVA). The green ANOVA diamonds have three horizontal lines. The middle, longer line indicates the mean value and the short lines indicate the upper and lower 95% confidence intervals. Because the confidence intervals overlap between different diamonds, this indicates no significant difference between the readings on different instruments. The right panel shows the results of a post-hoc Tukey-Kramer Honestly Significant Difference (HSD) test. The test compares each possible pair of results; when two circles overlap by half area, they are not significantly different.
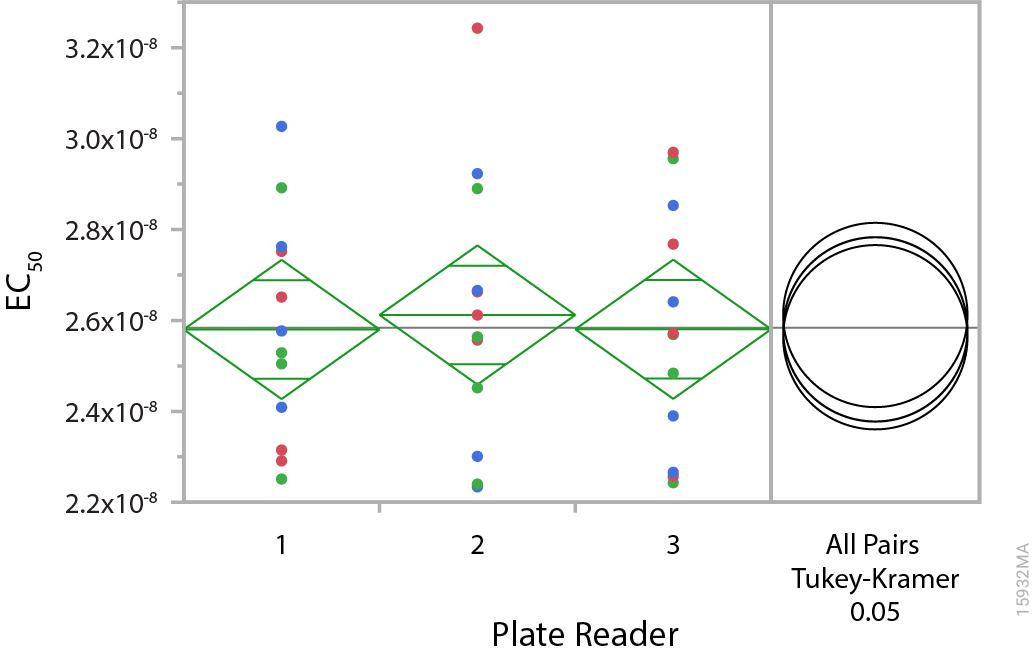
Figure 2. Comparison of EC50 measurements. Multiple ADCC Reporter Bioassays were performed over several days using multiple lots of effector and target (Raji) cell lines and an anti-CD20 antibody. Each bioassay plate was analyzed on three different GloMax® instruments and the results subjected to an analysis of variance (ANOVA). The green ANOVA diamonds have three horizontal lines. The middle, longer line indicates the mean value and the short lines indicate the upper and lower 95% confidence intervals. Because the confidence intervals overlap between different diamonds, this indicates no significant difference between the readings on different instruments. The right panel shows the results of a post-hoc Tukey-Kramer Honestly Significant Difference (HSD) test. The test compares each possible pair of results; when two circles overlap by half area, they are not significantly different.
Why Use GloMax® Instruments?
GloMax® plate readers can be used to measure a variety of detection modes, including luminescence, fluorescence and absorbance assays. Manuela and her team use luminescence primarily. They use assays such as our Bio-Glo™, Nano-Glo®, CellTiter-Glo™ and CytoTox-Glo™ products.
The Cell Manufacturing team uses the GloMax® Discover plate reader for a number of reasons. Not only is the instrument very intuitive, with an easy-to-use interface, it is also one of the most sensitive instruments on the market. Manuela’s team has multiple GloMax® plate readers and has also found that the data they get from reader to reader is very consistent. Plus, the high sensitivity and wide dynamic range make it easy to get good data even from samples with very low luminescence.
Designed and Optimized with Our Assays
Promega’s research and development team uses GloMax® plate readers to develop and optimize many of their cell health, metabolism and genetic reporter assays due to the instrument’s high sensitivity, wide dynamic range and low cross-talk. Those assay protocols are then built into the GloMax® Software as pre-programmed protocols to make it easier for customers (Figure 3). “The instrument gives us confidence we are getting good data even from samples with very low luminescence,” says Manuela. After measuring a plate, the results are displayed in a color heat map. "We love the results heat map because it makes it very easy to quickly identify any issues or trends with the data before we proceed to more involved calculations," Manuela says (Figure 4).
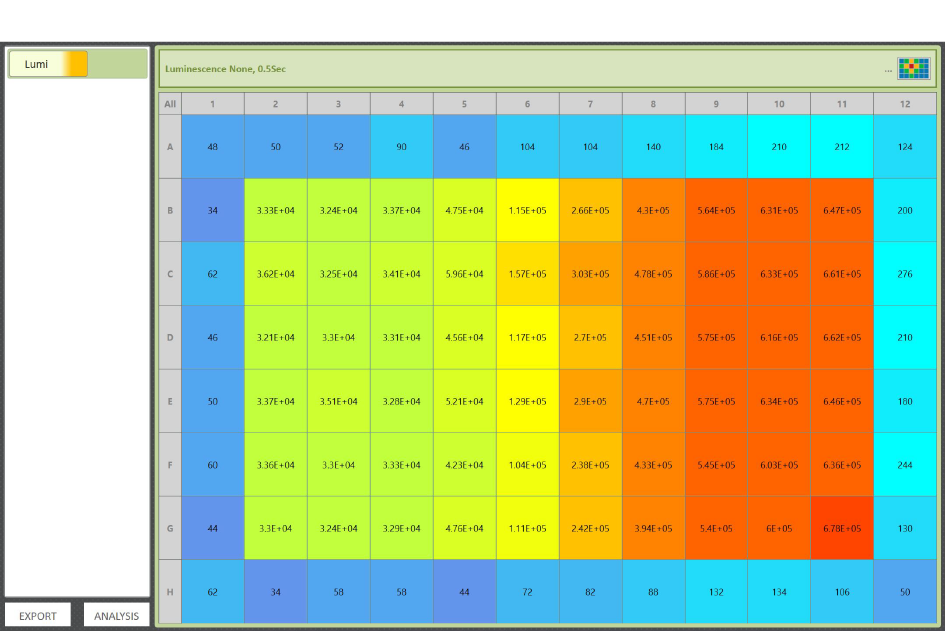
Another beneficial aspect about using GloMax® plate readers is that the instrument is designed to provide technical features for labs seeking part 11 compliance. The software includes user authentication and authorization, data integrity and protection, electronic signatures, and audit trails.
Summary
Manuela’s advice for anyone doing QC for cell-based assays is to use thaw-and-use cells because it will eliminate some of the variability from using cells from continuous culture. She also recommends to control critical steps in the bioassay like incubation times, and keep track of critical reagent lots. Having analysts that are specifically trained in using QC assays is certainly important, and of course, whenever possible, use a highly sensitive instrument like a GloMax® plate reader.
Try a GloMax® Instrument today!